554 JOURNAL OF COSMETIC SCIENCE
effect of makeup, making its efficient removal challenging with common surfactant-based
cleansers like soaps or oil-based cleansers.7
An effective alternative for skin cleansing consists of micellar waters. Micellar waters are
aqueous solutions containing surfactant molecules that form micelles, which are capable
of absorbing dirt and oil from the skin. These micelles consist of hydrophobic tails and
hydrophilic heads. When applied to the skin, the hydrophobic tails bind to oils and
impurities while the hydrophilic heads remain in the water, which allow the micelles to
encapsulate and lift away dirt and makeup effectively.8
Micellar waters are particularly gentle and suitable for sensitive skin because they clean
without the need for harsh scrubbing or rinsing. This makes them an ideal choice for those
with delicate skin conditions. Moreover, the nonionic surfactants used in some micellar
waters, such as glycerol esters, are designed to mimic the skin’s natural lipids, which ensure
a mild cleansing action that maintains the skin’s barrier without causing irritation.9
In addition to concerns about preserving the skin barrier, recent years have seen increased
attention on the impact of cosmetic products, particularly skin cleansers, on the skin
microbiota.10–12 Conventional soaps, for instance, can potentially disrupt the natural
microflora of the skin, thus resulting in imbalances within the microbiome.13 In addition
to the importance of developing makeup removal solutions that are microbiome-safe,
the adoption of strategies that adequately modulate the microbiome (such as the use of
prebiotics) has shown promise in restoring microbial balance and improving skin health.14
Prebiotic ingredients can exert positive effects on the host’s health by selectively stimulating
the growth of specific beneficial microbes.15
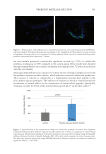
In this work, the effectiveness of a prebiotic aqueous micellar solution was tested specifically
for its ability to balance the skin’s microbiome while cleansing and removing makeup
without damaging the skin barrier.
MATERIALS AND METHODS
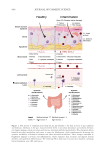
PREBIOTIC AQUEOUS MICELLAR SOLUTION
A commercially available bulk solution (Bio-Balance Water, Chemyunion Ltda., Sorocaba,
SP, Brazil) was tested for its efficacy in removing makeup and its impact on the facial skin
microbiome. This bulk solution contains inulin (medium concentration 3.0% w/w) and
xylitol (medium concentration 1.5% w/w) as prebiotic ingredients.
MAKEUP REMOVAL CAPACITY CLINICAL TEST
The study included 15 female volunteers, aged between 18 and 50, to whom commercially
available foundation (Base Liquida Vult HD Alta Cobertura – color 15, Vult, Mogi das
Cruzes, SP, Brazil), blush (Blush Vult – color 12, Vult, Mogi das Cruzes, SP, Brazil),
eyeshadow (Sombra Uno Vult MA01, Vult, Mogi das Cruzes, SP, Brazil), mascara (Máscara
para Cílios Vult 5UP! – black, Vult, Mogi das Cruzes, SP, Brazil), and lipstick (Batom Vult
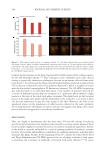
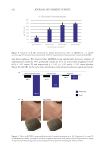
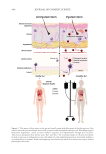
01 Cereja Cremoso, Vult, Mogi das Cruzes, SP, Brazil) were applied, as shown in Figure 1.
Front and side images were recorded with Visia® (Canfield Scientific, Inc., Parsippany, NJ,
USA) equipment before makeup application (T0), after makeup application (T
makeup ),and
effect of makeup, making its efficient removal challenging with common surfactant-based
cleansers like soaps or oil-based cleansers.7
An effective alternative for skin cleansing consists of micellar waters. Micellar waters are
aqueous solutions containing surfactant molecules that form micelles, which are capable
of absorbing dirt and oil from the skin. These micelles consist of hydrophobic tails and
hydrophilic heads. When applied to the skin, the hydrophobic tails bind to oils and
impurities while the hydrophilic heads remain in the water, which allow the micelles to
encapsulate and lift away dirt and makeup effectively.8
Micellar waters are particularly gentle and suitable for sensitive skin because they clean
without the need for harsh scrubbing or rinsing. This makes them an ideal choice for those
with delicate skin conditions. Moreover, the nonionic surfactants used in some micellar
waters, such as glycerol esters, are designed to mimic the skin’s natural lipids, which ensure
a mild cleansing action that maintains the skin’s barrier without causing irritation.9
In addition to concerns about preserving the skin barrier, recent years have seen increased
attention on the impact of cosmetic products, particularly skin cleansers, on the skin
microbiota.10–12 Conventional soaps, for instance, can potentially disrupt the natural
microflora of the skin, thus resulting in imbalances within the microbiome.13 In addition
to the importance of developing makeup removal solutions that are microbiome-safe,
the adoption of strategies that adequately modulate the microbiome (such as the use of
prebiotics) has shown promise in restoring microbial balance and improving skin health.14
Prebiotic ingredients can exert positive effects on the host’s health by selectively stimulating
the growth of specific beneficial microbes.15
In this work, the effectiveness of a prebiotic aqueous micellar solution was tested specifically
for its ability to balance the skin’s microbiome while cleansing and removing makeup
without damaging the skin barrier.
MATERIALS AND METHODS
PREBIOTIC AQUEOUS MICELLAR SOLUTION
A commercially available bulk solution (Bio-Balance Water, Chemyunion Ltda., Sorocaba,
SP, Brazil) was tested for its efficacy in removing makeup and its impact on the facial skin
microbiome. This bulk solution contains inulin (medium concentration 3.0% w/w) and
xylitol (medium concentration 1.5% w/w) as prebiotic ingredients.
MAKEUP REMOVAL CAPACITY CLINICAL TEST
The study included 15 female volunteers, aged between 18 and 50, to whom commercially
available foundation (Base Liquida Vult HD Alta Cobertura – color 15, Vult, Mogi das
Cruzes, SP, Brazil), blush (Blush Vult – color 12, Vult, Mogi das Cruzes, SP, Brazil),
eyeshadow (Sombra Uno Vult MA01, Vult, Mogi das Cruzes, SP, Brazil), mascara (Máscara
para Cílios Vult 5UP! – black, Vult, Mogi das Cruzes, SP, Brazil), and lipstick (Batom Vult
01 Cereja Cremoso, Vult, Mogi das Cruzes, SP, Brazil) were applied, as shown in Figure 1.
Front and side images were recorded with Visia® (Canfield Scientific, Inc., Parsippany, NJ,
USA) equipment before makeup application (T0), after makeup application (T
makeup ),and