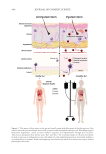
582 JOURNAL OF COSMETIC SCIENCE
Furthermore, lifestyle choices, including the use of cosmetics5 and dietary habits,6 have also
been shown to influence the skin’s microbial community. Numerous studies indicate that
the composition and balance of the skin microbiota can vary based on both external and
internal factors, such as skin integrity and physiological conditions.7 These conditions can
lead to various skin disorders, including atopic dermatitis,8 acne,9 psoriasis,10 and vitiligo.11
However, the origins of many skin diseases are complex and multifactorial. In particular
cases, such as SS syndrome, the precise role of microorganisms is still not fully understood,
although changes in the skin microbiota were already reported and suspected to contribute
to this condition.
For instance, we can document the findings of a first bacterial analysis by mass spectrometry
using MALDI-Biotyper of bacterial isolates collected and cultivated under aerobic
conditions. Isolates were collected on the forehead, cheek or scapula from 2 groups of
panelists of 3 men and 3 women (20–35 years old) having normal skin (NS) or SS evaluated
by lactic acid sting test (LAST). This well-recognized test allows diagnosis of the degree
of skin sensitivity and consists of the application of an aqueous lactic acid solution wiped
on the test site, while an inert control substance is used on the contralateral test site. After
application, a moderate to severe tingling sensation occurs and participants were asked to
grade the intensity of stinging using a 4-point scale (0 =no stinging 1 =slight stinging
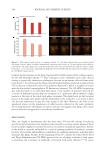
3 moderate stinging and ≥3 strong stinging). The results of the study showed that,
for both genders of the SS group, the relative abundance of Staphylococci decreased, and
Acinetobacter or Proteus were absent. Other bacterial variations were influenced by gender as
Pseudomonas and Bacillus that were absent in female SS. Acinetobacter and Proteus decreased
only in female SS, whereas Micrococcus increased only in male SS. Kocuria evolved in the
opposite sense between male and female SS, with an increase for female SS. However, in
this small cohort, no significant skin microbiome profile associated with the SS phenotype
was identified.12
In 2020, a larger study was performed on 42 Korean women (22–52 years old, cheek
area, sensitive selection by sodium lauryl sulfate patch test and LAST). The uncultivated
microbiome samples were analyzed for the first time using a more relevant but longer
method of microbiome identification at the species level based on analyses of hypervariable
bacterial 16S rRNA gene sequences or Internal Transcribed Spacer 1 (ITS1) fungal regions
(phylotype-based approaches using the closest type of strain assignment). The results
showed an equivalent diversity of skin bacteria between the 2 groups (NS and SS), but a
significant increase of Lactobacillus and Mucor racemosus, and a decrease in Malassezia restricta
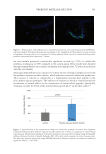
for SS.13 Another study was conducted using bacterial 16S rRNA gene sequencing on cheek
skin samples from 77 female panelists ranked in 3 groups according to LAST grading (31
NS samples with stinging test score 0 24 lightly SS with stinging test score 3, and 22 SS
with stinging test score ≥3). If skin bacterial diversity and richness were equivalent among
the 3 groups, on the genus level, there was no significant difference in the abundance
of Cutibacterium, while the abundance of Staphylococci in SS group was significantly lower
than in NS group. Interestingly, this study provided clear evidence that the abundance
of Staphylococcus epidermidis gradually decreased as skin sensitivity increased, whereas the
slight rise in Staphylococcus aureus observed was not statistically significant.14
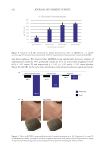
In 2021, the first microbiome study associated with a clinical evaluation was also performed
in China on 24 subjects with SS (face or chest, LAST-based recruitment) and 10 subjects
with NS (face).15 The cuticle moisture content, transepidermal water loss, facial skin sebum
Furthermore, lifestyle choices, including the use of cosmetics5 and dietary habits,6 have also
been shown to influence the skin’s microbial community. Numerous studies indicate that
the composition and balance of the skin microbiota can vary based on both external and
internal factors, such as skin integrity and physiological conditions.7 These conditions can
lead to various skin disorders, including atopic dermatitis,8 acne,9 psoriasis,10 and vitiligo.11
However, the origins of many skin diseases are complex and multifactorial. In particular
cases, such as SS syndrome, the precise role of microorganisms is still not fully understood,
although changes in the skin microbiota were already reported and suspected to contribute
to this condition.
For instance, we can document the findings of a first bacterial analysis by mass spectrometry
using MALDI-Biotyper of bacterial isolates collected and cultivated under aerobic
conditions. Isolates were collected on the forehead, cheek or scapula from 2 groups of
panelists of 3 men and 3 women (20–35 years old) having normal skin (NS) or SS evaluated
by lactic acid sting test (LAST). This well-recognized test allows diagnosis of the degree
of skin sensitivity and consists of the application of an aqueous lactic acid solution wiped
on the test site, while an inert control substance is used on the contralateral test site. After
application, a moderate to severe tingling sensation occurs and participants were asked to
grade the intensity of stinging using a 4-point scale (0 =no stinging 1 =slight stinging
3 moderate stinging and ≥3 strong stinging). The results of the study showed that,
for both genders of the SS group, the relative abundance of Staphylococci decreased, and
Acinetobacter or Proteus were absent. Other bacterial variations were influenced by gender as
Pseudomonas and Bacillus that were absent in female SS. Acinetobacter and Proteus decreased
only in female SS, whereas Micrococcus increased only in male SS. Kocuria evolved in the
opposite sense between male and female SS, with an increase for female SS. However, in
this small cohort, no significant skin microbiome profile associated with the SS phenotype
was identified.12
In 2020, a larger study was performed on 42 Korean women (22–52 years old, cheek
area, sensitive selection by sodium lauryl sulfate patch test and LAST). The uncultivated
microbiome samples were analyzed for the first time using a more relevant but longer
method of microbiome identification at the species level based on analyses of hypervariable
bacterial 16S rRNA gene sequences or Internal Transcribed Spacer 1 (ITS1) fungal regions
(phylotype-based approaches using the closest type of strain assignment). The results
showed an equivalent diversity of skin bacteria between the 2 groups (NS and SS), but a
significant increase of Lactobacillus and Mucor racemosus, and a decrease in Malassezia restricta
for SS.13 Another study was conducted using bacterial 16S rRNA gene sequencing on cheek
skin samples from 77 female panelists ranked in 3 groups according to LAST grading (31
NS samples with stinging test score 0 24 lightly SS with stinging test score 3, and 22 SS
with stinging test score ≥3). If skin bacterial diversity and richness were equivalent among
the 3 groups, on the genus level, there was no significant difference in the abundance
of Cutibacterium, while the abundance of Staphylococci in SS group was significantly lower
than in NS group. Interestingly, this study provided clear evidence that the abundance
of Staphylococcus epidermidis gradually decreased as skin sensitivity increased, whereas the
slight rise in Staphylococcus aureus observed was not statistically significant.14
In 2021, the first microbiome study associated with a clinical evaluation was also performed
in China on 24 subjects with SS (face or chest, LAST-based recruitment) and 10 subjects
with NS (face).15 The cuticle moisture content, transepidermal water loss, facial skin sebum