535 Foreword
microorganisms. As the research marches forward, Garlet details the exciting correlations
that have been identified between the signs of aging and fluctuations in specific populations
of microbes on the skin.
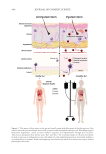
Schaefer et al. conclude this special skin microbiome issue with an elegant in-depth
elaboration of the current state of knowledge involving the interconnectivity of the gut
and the skin microbiomes. The so-called “gut-skin axis” has been described in some depth
elsewhere and more rarely the reverse “skin-gut axis” has been speculated upon. Here,
the authors comment on the bi-directionality of this important axis of communication
between members of the human microbiome. They also highlight how metagenomic
sequencing technology has been a major paradigm change from the early days where our
understanding of the skin microbiome was limited to what could be cultured from the
skin, and how these new research efforts are rapidly expanding our understanding of these
complex interactions and how they provide greater resolution of the microbes that are part
of these pathways.
Collectively, these articles contribute to the substantive advancement in our understanding
of not just the components of the skin microbiome, but the intricate interconnectedness of
this diverse community and its impacts on skin health.
Many unanswered questions and nebulous aspects remain, assuring exciting future
revelations to be made in this area of research. This frontier of skin microbiome topics
includes the unheralded members of the skin microbiota that are less often mentioned:
the fungi, protists, archaea, viruses, and others yet to be fully characterized. Indeed, the
discovery of a new group of acellular biological entities composed entirely of RNA called
“obelisks” was reported in early 2024, and they appear to be associated with the gut and
oral microbiomes, which potentially could extend to the skin. Also, pathogenic varieties
of prions, another acellular biological entity, have been found to be “seeded” into human
skin under certain conditions. The archaea certainly present an intriguing mystery given
that they are representative of the third cellular domain of life on Earth and are frequently
characterized as environmental extremophiles. Their precise role in skin health has yet to
be defined, but there appears to be an age component as they are detected at their highest
levels at early ages up to adolescence when there is a pronounced drop in their abundance
only to resurge again around the age of 60. Similarly, metagenomic sequencing efforts
have shown the viral component of the skin microbiota seems to be dominated by DNA-
based viruses, which could have deeper meaning or be an artifact of genomic sequencing
techniques that favor DNA genomes. These are just a few of the tantalizing questions yet to
be answered, which will certainly provoke discussions of how and whether the personal care
industry can capitalize on the resulting findings to design, develop, and launch new classes
of active ingredients that can improve skin health by modulating these microbial targets.
As we celebrate the 75th anniversary of the Journal of Cosmetic Science, it is our sincerest hope
that you will appreciate these intriguing reports relevant to the ongoing effort to expand
our knowledge and understanding of the skin microbiome. This is a subject area that
continues to demonstrate a profound relevance to the maintenance of healthy, youthful skin.
microorganisms. As the research marches forward, Garlet details the exciting correlations
that have been identified between the signs of aging and fluctuations in specific populations
of microbes on the skin.
Schaefer et al. conclude this special skin microbiome issue with an elegant in-depth
elaboration of the current state of knowledge involving the interconnectivity of the gut
and the skin microbiomes. The so-called “gut-skin axis” has been described in some depth
elsewhere and more rarely the reverse “skin-gut axis” has been speculated upon. Here,
the authors comment on the bi-directionality of this important axis of communication
between members of the human microbiome. They also highlight how metagenomic
sequencing technology has been a major paradigm change from the early days where our
understanding of the skin microbiome was limited to what could be cultured from the
skin, and how these new research efforts are rapidly expanding our understanding of these
complex interactions and how they provide greater resolution of the microbes that are part
of these pathways.
Collectively, these articles contribute to the substantive advancement in our understanding
of not just the components of the skin microbiome, but the intricate interconnectedness of
this diverse community and its impacts on skin health.
Many unanswered questions and nebulous aspects remain, assuring exciting future
revelations to be made in this area of research. This frontier of skin microbiome topics
includes the unheralded members of the skin microbiota that are less often mentioned:
the fungi, protists, archaea, viruses, and others yet to be fully characterized. Indeed, the
discovery of a new group of acellular biological entities composed entirely of RNA called
“obelisks” was reported in early 2024, and they appear to be associated with the gut and
oral microbiomes, which potentially could extend to the skin. Also, pathogenic varieties
of prions, another acellular biological entity, have been found to be “seeded” into human
skin under certain conditions. The archaea certainly present an intriguing mystery given
that they are representative of the third cellular domain of life on Earth and are frequently
characterized as environmental extremophiles. Their precise role in skin health has yet to
be defined, but there appears to be an age component as they are detected at their highest
levels at early ages up to adolescence when there is a pronounced drop in their abundance
only to resurge again around the age of 60. Similarly, metagenomic sequencing efforts
have shown the viral component of the skin microbiota seems to be dominated by DNA-
based viruses, which could have deeper meaning or be an artifact of genomic sequencing
techniques that favor DNA genomes. These are just a few of the tantalizing questions yet to
be answered, which will certainly provoke discussions of how and whether the personal care
industry can capitalize on the resulting findings to design, develop, and launch new classes
of active ingredients that can improve skin health by modulating these microbial targets.
As we celebrate the 75th anniversary of the Journal of Cosmetic Science, it is our sincerest hope
that you will appreciate these intriguing reports relevant to the ongoing effort to expand
our knowledge and understanding of the skin microbiome. This is a subject area that
continues to demonstrate a profound relevance to the maintenance of healthy, youthful skin.