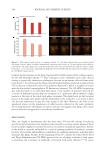
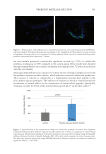
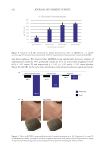
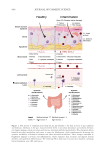
537 Modern Skincare
The largest constituent of this vast microbial community are the bacteria, which have
been implicated in many skin conditions. The skin bacteriome can vary considerably
depending upon which region of the skin is examined giving rise to the study of the
“skin biogeography” where specific groups and species of bacteria have been identified
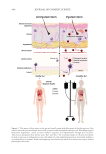
that prefer dry, moist, and sebaceous landscapes.1,5,6 For example, S. epidermidis has been
found to be abundant in moist regions, while the curiously anaerobic C. acnes exhibits a
preference for the oily, sebaceous sites. Indeed, C. acnes establishes colonies deep inside
the pilosebaceous units of the skin, where the bacteria construct stratified biofilms and
promote sebum production to be insulated from atmospheric oxygen impeding growth.7
Fluctuations in the homeostatic levels of these various microorganisms are referred to as
a “dysbiosis,” and many skin conditions have been attributed to diminished or excessive
growth of specific members of the microbiota. One such example is the case of increased
growth of Staphylococcus aureus, sometimes at the expense of S. epidermidis, contributing to
the incidence of atopic dermatitis.8,9 Similarly, although the precise molecular pathogenesis
remains to be delineated, and as the name suggests, C. acnes has been suggested to play a
key role in the development of acne vulgaris.3,10,11
Acne vulgaris is a fairly widespread skin disease afflicting adolescents and adults from
around the world inflicting both physiological and psychological stress on the individual.12–14
The classical model of C. acnes involvement in the pathogenesis of blemished skin suggests
that higher than normal growth of the bacteria leads to excessive sebum production and
induction of various inflammatory pathways.15 Other groups have reported evidence that
different strains of C. acnes are more likely to promote “acneic” conditions than others.11
Unfortunately, to date, the various proposed models do not account for all incidence of
acne in people, thus warranting further investigations into the microbiological component
of this skin disease.
Skin microbiome research has progressed from the early days where its scope was limited to
only those microbes that were “culturable” off the skin. The advent of 16S ribosomal RNA
(rRNA) sequencing allowed for “non-culturable” species to be identified and characterized.16
More recently, metagenomic sequencing has expanded our knowledge of the skin microbiota
beyond traditional bacteria to include archaea, fungi, protists, and viruses.1,17,18 Modern
skincare has sought to leverage these findings to deliver innovative topical solutions to
modulate and balance the skin microbiome for beneficial outcomes. However, given the
complexity of these interrelationships and the evidence that many of these microorganisms
make positive contributions to skin health, the need for precise, targeted interventions
becomes more apparent. To that end, the application of bacteriophage (literally “bacteria
consuming”) therapy, a technology dating back more than 100 years, provides an avenue
for skincare scientists to navigate that delicate balance.19–22 The precision of this approach
stems from the exquisite species-specificity (sometimes strain-specificity) exhibited by
naturally occurring bacteriophages for their bacterial targets. These bacterial viruses
recognize and attach to specific bacteria via unique molecules decorating their surfaces.23
For example, motifs within cell wall teichoic acids displayed on the thick peptidoglycan
(murein) cell walls of most Gram-positive bacteria are a common binding site for the
cognate bacteriophages of many skin-resident bacteria, including C. acnes and S. aureus.24
Once anchored to the surface of the bacterium, the bacteriophage will puncture the cell
wall and inject their nucleic acid genome (most commonly deoxyribonucleic acid [DNA])
inside, which thereupon will initiate a replication cycle that often terminates in the rupture
of the bacterial cell. This mode of bacteria destruction is what captured the attention
The largest constituent of this vast microbial community are the bacteria, which have
been implicated in many skin conditions. The skin bacteriome can vary considerably
depending upon which region of the skin is examined giving rise to the study of the
“skin biogeography” where specific groups and species of bacteria have been identified
that prefer dry, moist, and sebaceous landscapes.1,5,6 For example, S. epidermidis has been
found to be abundant in moist regions, while the curiously anaerobic C. acnes exhibits a
preference for the oily, sebaceous sites. Indeed, C. acnes establishes colonies deep inside
the pilosebaceous units of the skin, where the bacteria construct stratified biofilms and
promote sebum production to be insulated from atmospheric oxygen impeding growth.7
Fluctuations in the homeostatic levels of these various microorganisms are referred to as
a “dysbiosis,” and many skin conditions have been attributed to diminished or excessive
growth of specific members of the microbiota. One such example is the case of increased
growth of Staphylococcus aureus, sometimes at the expense of S. epidermidis, contributing to
the incidence of atopic dermatitis.8,9 Similarly, although the precise molecular pathogenesis
remains to be delineated, and as the name suggests, C. acnes has been suggested to play a
key role in the development of acne vulgaris.3,10,11
Acne vulgaris is a fairly widespread skin disease afflicting adolescents and adults from
around the world inflicting both physiological and psychological stress on the individual.12–14
The classical model of C. acnes involvement in the pathogenesis of blemished skin suggests
that higher than normal growth of the bacteria leads to excessive sebum production and
induction of various inflammatory pathways.15 Other groups have reported evidence that
different strains of C. acnes are more likely to promote “acneic” conditions than others.11
Unfortunately, to date, the various proposed models do not account for all incidence of
acne in people, thus warranting further investigations into the microbiological component
of this skin disease.
Skin microbiome research has progressed from the early days where its scope was limited to
only those microbes that were “culturable” off the skin. The advent of 16S ribosomal RNA
(rRNA) sequencing allowed for “non-culturable” species to be identified and characterized.16
More recently, metagenomic sequencing has expanded our knowledge of the skin microbiota
beyond traditional bacteria to include archaea, fungi, protists, and viruses.1,17,18 Modern
skincare has sought to leverage these findings to deliver innovative topical solutions to
modulate and balance the skin microbiome for beneficial outcomes. However, given the
complexity of these interrelationships and the evidence that many of these microorganisms
make positive contributions to skin health, the need for precise, targeted interventions
becomes more apparent. To that end, the application of bacteriophage (literally “bacteria
consuming”) therapy, a technology dating back more than 100 years, provides an avenue
for skincare scientists to navigate that delicate balance.19–22 The precision of this approach
stems from the exquisite species-specificity (sometimes strain-specificity) exhibited by
naturally occurring bacteriophages for their bacterial targets. These bacterial viruses
recognize and attach to specific bacteria via unique molecules decorating their surfaces.23
For example, motifs within cell wall teichoic acids displayed on the thick peptidoglycan
(murein) cell walls of most Gram-positive bacteria are a common binding site for the
cognate bacteriophages of many skin-resident bacteria, including C. acnes and S. aureus.24
Once anchored to the surface of the bacterium, the bacteriophage will puncture the cell
wall and inject their nucleic acid genome (most commonly deoxyribonucleic acid [DNA])
inside, which thereupon will initiate a replication cycle that often terminates in the rupture
of the bacterial cell. This mode of bacteria destruction is what captured the attention