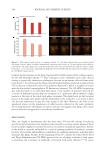
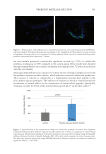
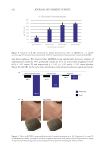
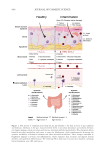
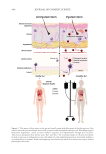
613 Pentapeptide to Control Acne
either palmitic acid (1.56–3.12 ppm), isolated peptide (KTSKS 4.525–9.05 ppm), or with
a blend of both compounds, all at an equivalent concentration of pKTSKS, to evaluate the
effects of each compound on C. acnes growth over time. The same previously described
culture conditions were used.
S. epidermidis (DSM 28764, Germany) was routinely cultivated in TSY media: 30g/L
tryptic soy broth (Biomérieux, Craponne, France) and 3g/L yeast extract (Organotechnie,
La Courneuve, France). For growth studies, bacteria were seeded at 7 × 107 CFU/mL in 96
well plates and growth were monitored using a Bioscreen C (Bioscreen, Finland).
ADHESION AND BIOFILM ASSAYS
C. acnes RT-1 adhesion and biofilm formation were evaluated on the surface of plastic plates.
106 CFU/mL were seeded in liquid medium (Trypticase soy) ± pKTSKS (6–12 ppm). In
absence of pKTSKS, its solvent (DMSO) was used as a control. For adhesion, pKTSKS was
added to culture medium immediately after seeding. Cell culture medium was removed
after 3 days, and adherent cells were estimated using method derived from experiments
described by Merritt et al.,22 at BioEC and Cergy Pontoise University Laboratories (Cergy,
France). Briefly, adherent cells were rinsed and stained with crystal violet for 20 minutes.
Unbound colorant was discarded, and cells rinsed with water, then colorant was extracted
in 33% acetic acid and variations of OD
600nm were evaluated with a spectrophotometer. For
biofilm formation study, the same protocol was used but pKTSKS was added to culture
medium 8 hours after seeding of cells.
ASSAYS ON HUMAN KERATINOCYTES
HaCaT cells, spontaneously immortalized human keratinocyte line,23 were obtained from
DKFZ (Heidelberg, Germany) and cultured with 5% CO
2 at 37°C and 90% humidity
atmosphere in Dulbecco’s Modified Eagle Medium (DMEM Gibco, Thermo Fischer
Scientific, Illkirch-Graffenstaden, France) supplemented with 0.09 mM Ca²+ (Sigma
Aldrich, Germany), 4 mM glutamine (Gibco), 1 mM sodium pyruvate (Gibco), 100 IU/
mL penicillin-streptomycin (Gibco), and 1 µg/mL Amphotericin B (Gibco). For routine
cultures, medium was supplemented with 10% heat-inactivated fetal bovine serum (FBS,
Gibco). Media was renewed every 48–72 hours to allow cell growth. For experiments, cells
were seeded between 8 – 16 × 104 cells/cm² and cultured into DMEM with 10% FBS
until confluency was reached. Cells were then switched to DMEM without antibiotics nor
antifungal compounds to guarantee C. acnes survival. For interleukin-6 (IL-6), interleukin
1-receptor antagonist (IL-1Ra), and hyaluronic acid, cells were treated with pKTSKS
peptide (6–12 ppm) or its solvent (0.1% DMSO, Sigma Aldrich, Germany) for 3 days. After
the first 24 hours of treatment, living planktonic C. acnes cells (100 bacteria: 1 keratinocyte,
for IL-6 and -1Ra only) were added for 48 hours. Then, cell culture media were collected
for cytokine and hyaluronic acid measurements using ELISA methods (IL-6 and IL1-RA:
R&D Systems Hyaluronic acid: Corgenix 029-001) while cell viability was evaluated using
nucleus labeling with Hoechst 33258 method (Sigma Aldrich, Germany).24 For IL-6, plates
were coated over night at 4°C with an anti-IL-6 antibody. For IL1-RA and hyaluronic acid,
plates were precoated. Briefly samples (or standard) were added to the plate and incubated
either palmitic acid (1.56–3.12 ppm), isolated peptide (KTSKS 4.525–9.05 ppm), or with
a blend of both compounds, all at an equivalent concentration of pKTSKS, to evaluate the
effects of each compound on C. acnes growth over time. The same previously described
culture conditions were used.
S. epidermidis (DSM 28764, Germany) was routinely cultivated in TSY media: 30g/L
tryptic soy broth (Biomérieux, Craponne, France) and 3g/L yeast extract (Organotechnie,
La Courneuve, France). For growth studies, bacteria were seeded at 7 × 107 CFU/mL in 96
well plates and growth were monitored using a Bioscreen C (Bioscreen, Finland).
ADHESION AND BIOFILM ASSAYS
C. acnes RT-1 adhesion and biofilm formation were evaluated on the surface of plastic plates.
106 CFU/mL were seeded in liquid medium (Trypticase soy) ± pKTSKS (6–12 ppm). In
absence of pKTSKS, its solvent (DMSO) was used as a control. For adhesion, pKTSKS was
added to culture medium immediately after seeding. Cell culture medium was removed
after 3 days, and adherent cells were estimated using method derived from experiments
described by Merritt et al.,22 at BioEC and Cergy Pontoise University Laboratories (Cergy,
France). Briefly, adherent cells were rinsed and stained with crystal violet for 20 minutes.
Unbound colorant was discarded, and cells rinsed with water, then colorant was extracted
in 33% acetic acid and variations of OD
600nm were evaluated with a spectrophotometer. For
biofilm formation study, the same protocol was used but pKTSKS was added to culture
medium 8 hours after seeding of cells.
ASSAYS ON HUMAN KERATINOCYTES
HaCaT cells, spontaneously immortalized human keratinocyte line,23 were obtained from
DKFZ (Heidelberg, Germany) and cultured with 5% CO
2 at 37°C and 90% humidity
atmosphere in Dulbecco’s Modified Eagle Medium (DMEM Gibco, Thermo Fischer
Scientific, Illkirch-Graffenstaden, France) supplemented with 0.09 mM Ca²+ (Sigma
Aldrich, Germany), 4 mM glutamine (Gibco), 1 mM sodium pyruvate (Gibco), 100 IU/
mL penicillin-streptomycin (Gibco), and 1 µg/mL Amphotericin B (Gibco). For routine
cultures, medium was supplemented with 10% heat-inactivated fetal bovine serum (FBS,
Gibco). Media was renewed every 48–72 hours to allow cell growth. For experiments, cells
were seeded between 8 – 16 × 104 cells/cm² and cultured into DMEM with 10% FBS
until confluency was reached. Cells were then switched to DMEM without antibiotics nor
antifungal compounds to guarantee C. acnes survival. For interleukin-6 (IL-6), interleukin
1-receptor antagonist (IL-1Ra), and hyaluronic acid, cells were treated with pKTSKS
peptide (6–12 ppm) or its solvent (0.1% DMSO, Sigma Aldrich, Germany) for 3 days. After
the first 24 hours of treatment, living planktonic C. acnes cells (100 bacteria: 1 keratinocyte,
for IL-6 and -1Ra only) were added for 48 hours. Then, cell culture media were collected
for cytokine and hyaluronic acid measurements using ELISA methods (IL-6 and IL1-RA:
R&D Systems Hyaluronic acid: Corgenix 029-001) while cell viability was evaluated using
nucleus labeling with Hoechst 33258 method (Sigma Aldrich, Germany).24 For IL-6, plates
were coated over night at 4°C with an anti-IL-6 antibody. For IL1-RA and hyaluronic acid,
plates were precoated. Briefly samples (or standard) were added to the plate and incubated