615 Pentapeptide to Control Acne
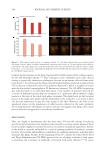
biotin-conjugated anti-collagen-IV antibody for 1 hour, followed by the substrate solution
(containing streptavidin-phycoerythrin conjugate) for 15 minutes at room temperature. The
reaction was then stopped, and absorbance was measured at 450 nm using a microplate
reader.
EQUIVALENT SKIN MODEL
Human Living Skin Equivalents (LSE) were produced in-house (adapted of method described
by Asselineau et al.25). Briefly, for equivalent-dermis, NHF were seeded into collagen-I
matrix (Jacques Boy, France) in 6-well plate inserts (Falcon™, New York, USA) and kept
emerged for 48 hours to allow dermal contraction. Human keratinocytes (NHK, CellnTec)
were seeded on top of the equivalent dermis. Models were cultivated for 7 days in immersion
media, followed by 7 days of air-liquid interphase to obtain a stratified epidermis. 9 ppm of
pKTSKS or its solvent (0.1% DMSO) were applied on top of the epidermis. After 48 hours
of treatment, LSE were embedded in freezing media (Leica, UK) and snap-frozen in liquid
nitrogen. 5–7 µm sections were prepared using a cryostat (CM15105, Leica, Germany). LSE
histology was evaluated by H&E (Sigma Aldrich, Germany) staining. Thickness of stratum
corneum was quantified on pictures captured under the microscope (Eclipse TI) using NIS
Element Software (both Nikon, Japan).
CLINICAL TRIALS
Two single-blind studies were performed on volunteers with acne-prone skin and acne scars.
The first study conducted on a panel of 30 volunteers made of 7 men and 23 women aged
between 18–39 years old with a mean age of 24 years old and a phototype III, was aimed
to measure pKTSKS effects on C. acnes and S. epidermidis populations. The second study
dedicated to topographic measurements was included a panel of 18 volunteers comprising
one man and 17 women aged from 20–58 years old with a mean age of 32, phototype II–IV
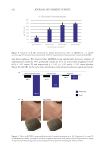
to evaluate the peptide’s effects on skin texture and scar improvement. Creams containing
12 ppm pKTSKS or placebo were formulated (see the formula in Figure 1). Volunteers
applied placebo on one cheek and pKTSKS-cream on the other, twice a day for up to 2
months. To study effects on C. acnes and S. epidermidis populations, RT-qPCR (real time
quantitative polymer chain reaction) analysis was performed on skin microbiota sampled
from 30 volunteers, before and after 1 month treatment. In the topographic variation
study, acquisitions were taken on 18 volunteers before and after 2 months treatment (this
relatively low number of volunteers was due to many dropouts linked to the Covid-19
pandemic). An Antera3D camera was used for analysis of various topographic parameters
such as inflammatory marks, pockmark, and skin roughness.
STATISTICS
ANOVA was used to determine whether there was any significant difference between
the means of two or more independent groups. Difference between two means with
similar variances was performed with Student’s t-test. A p-value p 0.05 or p 0.01 was
considered statistically significant.
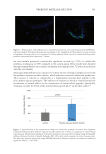
biotin-conjugated anti-collagen-IV antibody for 1 hour, followed by the substrate solution
(containing streptavidin-phycoerythrin conjugate) for 15 minutes at room temperature. The
reaction was then stopped, and absorbance was measured at 450 nm using a microplate
reader.
EQUIVALENT SKIN MODEL
Human Living Skin Equivalents (LSE) were produced in-house (adapted of method described
by Asselineau et al.25). Briefly, for equivalent-dermis, NHF were seeded into collagen-I
matrix (Jacques Boy, France) in 6-well plate inserts (Falcon™, New York, USA) and kept
emerged for 48 hours to allow dermal contraction. Human keratinocytes (NHK, CellnTec)
were seeded on top of the equivalent dermis. Models were cultivated for 7 days in immersion
media, followed by 7 days of air-liquid interphase to obtain a stratified epidermis. 9 ppm of
pKTSKS or its solvent (0.1% DMSO) were applied on top of the epidermis. After 48 hours
of treatment, LSE were embedded in freezing media (Leica, UK) and snap-frozen in liquid
nitrogen. 5–7 µm sections were prepared using a cryostat (CM15105, Leica, Germany). LSE
histology was evaluated by H&E (Sigma Aldrich, Germany) staining. Thickness of stratum
corneum was quantified on pictures captured under the microscope (Eclipse TI) using NIS
Element Software (both Nikon, Japan).
CLINICAL TRIALS
Two single-blind studies were performed on volunteers with acne-prone skin and acne scars.
The first study conducted on a panel of 30 volunteers made of 7 men and 23 women aged
between 18–39 years old with a mean age of 24 years old and a phototype III, was aimed
to measure pKTSKS effects on C. acnes and S. epidermidis populations. The second study
dedicated to topographic measurements was included a panel of 18 volunteers comprising
one man and 17 women aged from 20–58 years old with a mean age of 32, phototype II–IV
to evaluate the peptide’s effects on skin texture and scar improvement. Creams containing
12 ppm pKTSKS or placebo were formulated (see the formula in Figure 1). Volunteers
applied placebo on one cheek and pKTSKS-cream on the other, twice a day for up to 2
months. To study effects on C. acnes and S. epidermidis populations, RT-qPCR (real time
quantitative polymer chain reaction) analysis was performed on skin microbiota sampled
from 30 volunteers, before and after 1 month treatment. In the topographic variation
study, acquisitions were taken on 18 volunteers before and after 2 months treatment (this
relatively low number of volunteers was due to many dropouts linked to the Covid-19
pandemic). An Antera3D camera was used for analysis of various topographic parameters
such as inflammatory marks, pockmark, and skin roughness.
STATISTICS
ANOVA was used to determine whether there was any significant difference between
the means of two or more independent groups. Difference between two means with
similar variances was performed with Student’s t-test. A p-value p 0.05 or p 0.01 was
considered statistically significant.