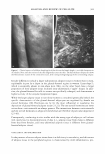
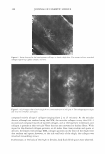
COSMECEUTICAL PROPER TIES OF LEV AN 401 Figure 2. TEM micrograph of levan showing self-formed nanoparticles. concentrations, ranging from 10 wt% to 50 wt%. Turbidity and precipitation were observed in order to determine the storage stability at room temperature, 5°C, and 40°C for a total of 45 days. As a result of these observations, we determined the solubility of the levan solution in ethanol to be so excellent that we were completely unable to observe any precipitation, and the solution was found not to be turbid (Table II). MEASUREMENT OF MOISTURIZING EFFECT To evaluate the moisturizing effects exerted by levan, we measured transepidermal water loss (TEWL) and moisture content with a Corneometer CM825. The TEWL results of the levan applied to the skin were then compared with the results of 0.2% hyaluronic acid and distilled water (Figure 3). Our statistical analysis of this data revealed a significant decrease in water loss on the skin as the result of the application of levan and hyaluronic acid, as opposed to when distilled water was applied. We then measured the moisture content of 0.2% levan and 0.2% hyaluronic acid with the Corneometer CM825 Table II Stability Test of Levan in Aqueous Ethanol 1 Ethanol (%)2 10 20 30 40 50 7 stable stable stable stable stable 15 stable stable stable stable stable 1 Levan was used at a concentration of 5 % on a solid basis. 2 Ethanol(%) was expressed by weight of ethanol in water. 3 Kept at room temperature, 5°, and 40°C for 45 days. Period (days)3 30 45 stable stable stable stable stable stable stable stable stable stable
402 110 100 � 90 C .! C 8 80 .! 70 60 50 JOURNAL OF COSMETIC SCIENCE 0 5 10 -+- Levan -o-- Hyaluronic acid 1% 15 Time (hr) 20 25 30 35 Figure 3. Transepidermal water loss (TEWL) of levan and hyaluronic acid (HA) as a function of time. The data are expressed as mean values (± standard deviations) of five experiments. (Figure 4). We observed a trend almost identical to the one listed above. On the basis of these two results, it appears that levan exhibits a substantial moisturizing effect, although not as substantial as that determined for hyaluronic acid. CYTOTOXICITY ASSAY Data regarding cell cytotox1e1ty and proliferation effects as determined by the MTT assay are shown in Figures 5 and 6, respectively. Levan exhibited no cytotoxicity in the human fibroblast cell line, up to a relatively high concentration (100 µg/ml) (Figure 5). Moreover, levan exhibited a considerable cell proliferation effect in the keratinocyte cell 12 -+- Levan 0.2% 10 -o- Hyaluronic acid 0.2% G 8 1 6 G 0 G 4 0 2 0 0 5 10 15 20 25 30 35 Time (min) Figure 4. Moisturizing effect measured by Corneometer CM 825. The data are expressed as mean values (± standard deviations) of five experiments.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)