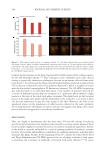
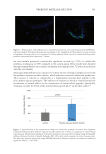
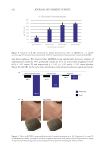
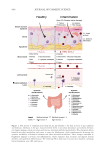
638 JOURNAL OF COSMETIC SCIENCE
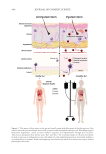
by two anaerobic bacterial phyla: Bacteroidetes and Firmicutes.32 Key genera involved in this
production include Bifidobacterium, Eubacterium, Lactobacillus and Prevotella.33 Roseburia, a
genus within the Lachnospiraceae family, is a key producer of butyrate in the gut.27 These
SCFAs serve critical functions including blood pressure regulation, post-infarction heart
healing, anti-inflammation and gut barrier function by serving as an energy source for
intestinal epithelial cells.34
Varied amounts of SCFAs enter the bloodstream, influenced by factors such as dietary fiber
intake, microbial fermentation rates and the extent of colon absorption. Once absorbed,
SCFAs interact with G-coupled-protein receptors such as free fatty acid receptor 2 (FFAR2,
also known as GPR43), the free fatty acid receptor 3 (FFAR3, also known as GPR41) and
the free fatty acid receptor GPR109a. These are expressed in tissues including adipocytes,
intestinal epithelial cells, pancreatic beta-cells, the spleen and immune cells such as M2
macrophages, neutrophils, eosinophils and mast cells.32,35 Regarding the skin, SCFAs can
dampen immunoglobulin E (IgE) allergic inflammatory responses, increase cholesterol and
ceramide concentrations in the stratum corneum, repress transepidermal water loss for the
maintenance of the epidermal barrier and inhibit histone deacetylation of keratinocytes to
further suppress inflammation.31 Therefore, SCFAs may directly affect the skin or alter the
skin’s commensal bacteria. However, further research is needed to determine the clinically
significant amount of SCFAs that must reach the bloodstream to impact the skin.30
Other gut microbial metabolites associated with skin conditions include tryptophan
metabolites.36 In the gut, commensal colon-inhabiting microbes like Clostridium, Bacteroides,
Bifidobacterium and Lactobacillus convert tryptophan to indole and its derivatives, such as
indole-3-aldehyde (IAId), indole-3-lactic acid (ILA), indole-3-propionic acid (IPA), indole-
3-acetic acid (IAA), indole-3-acrylic acid (IA) and tryptamine, all of which play key roles
in maintaining intestinal immune balance and barrier function.37,38 IPA in specific has
demonstrated the ability of enhancing intestinal barrier function in vitro and murine
models.39 Furthermore, products of this indole pathway, like indole-3-carbaldehyde (I3A),
can activate the aryl hydrocarbon receptor (AhR), which is widely expressed in skin cells
like fibroblasts, keratinocytes, Langerhans cells, melanocytes, sebocytes, mast cells and
lymphocytes.40–45 The effects of AhR stimulation are dependent on the dose and the specific
ligand, influencing different transcriptional pathways and inducing various biological
responses.40 AhR activation in the skin has been shown to enhance the production of key
skin barrier proteins, improving skin hydration and reducing water loss, while also causing
the upregulation of metalloproteinases and suppression of type I collagen and fibronectin
expression, thereby improving wound healing and decreasing scar formation.46 In addition,
another bacterial metabolite of tryptophan is indole pyruvate (IPyr) and it exerts a protective
effect on keratinocytes exposed to UVB.47
Finally, amine derivatives—such as trimethylamine (TMA) and trimethylamine N-oxide
(TMAO) —produced by the gut microbiome have been associated with skin health.36 These
amine derivatives are products of the intestinal microbiome’s degradation of quaternary
amine group-containing molecules, such as choline, L-carnitine or phosphatidylcholine
found in eggs, liver and dairy.48 TMA is generated by bacterial genera such as Clostridium,
Proteus, Shigella and Enterobacter and is then transported to the liver where it is oxidized
to produce TMAO.49 Further research is required to fully understand the implications of
TMA and TMAO in skin health. However, in general, elevated levels of these metabolites
have been associated with skin disorders such as psoriasis, systemic lupus erythematosus
and hidradenitis suppurativa.49–51
by two anaerobic bacterial phyla: Bacteroidetes and Firmicutes.32 Key genera involved in this
production include Bifidobacterium, Eubacterium, Lactobacillus and Prevotella.33 Roseburia, a
genus within the Lachnospiraceae family, is a key producer of butyrate in the gut.27 These
SCFAs serve critical functions including blood pressure regulation, post-infarction heart
healing, anti-inflammation and gut barrier function by serving as an energy source for
intestinal epithelial cells.34
Varied amounts of SCFAs enter the bloodstream, influenced by factors such as dietary fiber
intake, microbial fermentation rates and the extent of colon absorption. Once absorbed,
SCFAs interact with G-coupled-protein receptors such as free fatty acid receptor 2 (FFAR2,
also known as GPR43), the free fatty acid receptor 3 (FFAR3, also known as GPR41) and
the free fatty acid receptor GPR109a. These are expressed in tissues including adipocytes,
intestinal epithelial cells, pancreatic beta-cells, the spleen and immune cells such as M2
macrophages, neutrophils, eosinophils and mast cells.32,35 Regarding the skin, SCFAs can
dampen immunoglobulin E (IgE) allergic inflammatory responses, increase cholesterol and
ceramide concentrations in the stratum corneum, repress transepidermal water loss for the
maintenance of the epidermal barrier and inhibit histone deacetylation of keratinocytes to
further suppress inflammation.31 Therefore, SCFAs may directly affect the skin or alter the
skin’s commensal bacteria. However, further research is needed to determine the clinically
significant amount of SCFAs that must reach the bloodstream to impact the skin.30
Other gut microbial metabolites associated with skin conditions include tryptophan
metabolites.36 In the gut, commensal colon-inhabiting microbes like Clostridium, Bacteroides,
Bifidobacterium and Lactobacillus convert tryptophan to indole and its derivatives, such as
indole-3-aldehyde (IAId), indole-3-lactic acid (ILA), indole-3-propionic acid (IPA), indole-
3-acetic acid (IAA), indole-3-acrylic acid (IA) and tryptamine, all of which play key roles
in maintaining intestinal immune balance and barrier function.37,38 IPA in specific has
demonstrated the ability of enhancing intestinal barrier function in vitro and murine
models.39 Furthermore, products of this indole pathway, like indole-3-carbaldehyde (I3A),
can activate the aryl hydrocarbon receptor (AhR), which is widely expressed in skin cells
like fibroblasts, keratinocytes, Langerhans cells, melanocytes, sebocytes, mast cells and
lymphocytes.40–45 The effects of AhR stimulation are dependent on the dose and the specific
ligand, influencing different transcriptional pathways and inducing various biological
responses.40 AhR activation in the skin has been shown to enhance the production of key
skin barrier proteins, improving skin hydration and reducing water loss, while also causing
the upregulation of metalloproteinases and suppression of type I collagen and fibronectin
expression, thereby improving wound healing and decreasing scar formation.46 In addition,
another bacterial metabolite of tryptophan is indole pyruvate (IPyr) and it exerts a protective
effect on keratinocytes exposed to UVB.47
Finally, amine derivatives—such as trimethylamine (TMA) and trimethylamine N-oxide
(TMAO) —produced by the gut microbiome have been associated with skin health.36 These
amine derivatives are products of the intestinal microbiome’s degradation of quaternary
amine group-containing molecules, such as choline, L-carnitine or phosphatidylcholine
found in eggs, liver and dairy.48 TMA is generated by bacterial genera such as Clostridium,
Proteus, Shigella and Enterobacter and is then transported to the liver where it is oxidized
to produce TMAO.49 Further research is required to fully understand the implications of
TMA and TMAO in skin health. However, in general, elevated levels of these metabolites
have been associated with skin disorders such as psoriasis, systemic lupus erythematosus
and hidradenitis suppurativa.49–51