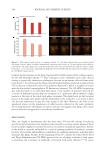
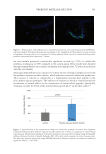
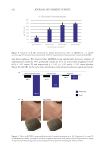
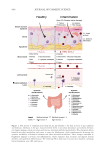
652 JOURNAL OF COSMETIC SCIENCE
(22) Sun J, Xia Y. Pretreating and normalizing metabolomics data for statistical analysis. Genes Dis. July
2024 11(3):100979. doi:10.1016/j.gendis.2023.04.018
(23) Walczak-Skierska J, Monedeiro F, Maślak E, Złoch M. Lipidomics characterization of the microbiome
in people with diabetic foot infection using MALDI-TOF MS. Anal Chem. 2023 95(44):16251–16262.
doi:10.1021/acs.analchem.3c03071
(24) Holčapek M, Liebisch G, Ekroos K. Lipidomic analysis. Anal Chem. 2018 90(7):4249–4257. doi:10.1021/
acs.analchem.7b05395
(25) Sinha S, Lin G, Ferenczi K. The skin microbiome and the gut-skin axis. Clin Dermatol. 2021 39(5):829–839.
doi:10.1016/j.clindermatol.2021.08.021
(26) Salem I, Ramser A, Isham N, Ghannoum MA. The gut microbiome as a major regulator of the gut-skin
axis. Front Microbiol. July 2018 9:1459. doi:10.3389/fmicb.2018.01459
(27) Greene AK, Nelson AM. The gut microbiome in melanoma: A piece of a complex puzzle. JAMA
Dermatol. 2023 159(10):1044–1046. doi:10.1001/jamadermatol.2023.2952
(28) Maguire M, Maguire G. The role of microbiota, and probiotics and prebiotics in skin health. Arch
Dermatol Res. 2017 309(6):411–421. doi:10.1007/s00403-017-1750-3
(29) Codoñer FM, Ramírez-Bosca A, Climent E, et al. Gut microbial composition in patients with psoriasis.
Sci Rep. February 2018 8(1):3812. doi:10.1038/s41598-018-22125-y
(30) O’Neill CA, Monteleone G, McLaughlin JT, Paus R. The gut-skin axis in health and disease: A paradigm
with therapeutic implications. BioEssays. 2016 38(11):1167–1176. doi:10.1002/bies.201600008
(31) Stec A, Sikora M, Maciejewska M, et al. Bacterial metabolites: A link between gut microbiota and
dermatological diseases. Int J Mol Sci. February 2023 24(4). doi:10.3390/ijms24043494
(32) Tan J, McKenzie C, Potamitis M, et al. Insights into the gut-skin axis: implications for skin disorders.
Clin Microbiol Rev. 2018 31:e00101–18.
(33) Cummings JH, Macfarlane GT. Role of intestinal bacteria in nutrient metabolism. Clin Nutr.
1997 16:3–11.
(34) Chakraborty P, Banerjee D, Majumder P, Sarkar J. Gut microbiota nexus: exploring the interactions
with the brain, heart, lungs, and skin axes and their effects on health, Med. Microecol. March 2024 20.
(35) Kimura I, Ichimura A, Ohue-Kitano R, Igarashi M. Free fatty acid receptors in health and disease.
Physiol Rev. 2020 100(1):171–210. doi:10.1152/physrev.00041.2018
(36) Jiminez V, Yusuf N. Bacterial metabolites and inflammatory skin diseases. Metabolites. August 2023 13(8).
doi:10.3390/metabo13080952
(37) Huang Y, Liu L, Hao Z, et al. Potential roles of gut microbial tryptophan metabolites in the complex
pathogenesis of acne vulgaris. Front Microbiol. July 2022 13:942027. doi:10.3389/fmicb.2022.942027
(38) Roager HM, Licht TR. Microbial tryptophan catabolites in health and disease. Nat Commun. August
2018 9(1):3294. doi:10.1038/s41467-018-05470-4
(39) Jennis M, Cavanaugh CR, Leo GC, Mabus JR, Lenhard J, Hornby PJ. Microbiota-derived tryptophan
indoles increase after gastric bypass surgery and reduce intestinal permeability in vitro and in vivo.
Neurogastroenterol Motil. August 2018 30(2). doi:10.1111/nmo.13178
(40) Esser C, Rannug A. The aryl hydrocarbon receptor in barrier organ physiology, immunology, and
toxicology. Pharmacol Rev. 2015 67(2):259–279. doi:10.1124/pr.114.009001
(41) Yu J, Luo Y, Zhu Z, et al. A tryptophan metabolite of the skin microbiota attenuates inflammation
in patients with atopic dermatitis through the aryl hydrocarbon receptor. J Allergy Clin Immunol.
2019 143:2108–2119.e12.
(42) Liu X, Zhang X, Zhang J, et al. Activation of aryl hydrocarbon receptor in Langerhans cells by a microbial
metabolite of tryptophan negatively regulates skin inflammation. J Dermatol Sci. 2020 100(3):192–200.
doi:10.1016/j.jdermsci.2020.10.004
(43) Jux B, Kadow S, Luecke S, Rannug A, Krutmann J, Esser C. The aryl hydrocarbon receptor mediates
UVB radiation-induced skin tanning. J Invest Dermatol. 2011 131(1):203–210. doi:10.1038/jid.2010.269
(22) Sun J, Xia Y. Pretreating and normalizing metabolomics data for statistical analysis. Genes Dis. July
2024 11(3):100979. doi:10.1016/j.gendis.2023.04.018
(23) Walczak-Skierska J, Monedeiro F, Maślak E, Złoch M. Lipidomics characterization of the microbiome
in people with diabetic foot infection using MALDI-TOF MS. Anal Chem. 2023 95(44):16251–16262.
doi:10.1021/acs.analchem.3c03071
(24) Holčapek M, Liebisch G, Ekroos K. Lipidomic analysis. Anal Chem. 2018 90(7):4249–4257. doi:10.1021/
acs.analchem.7b05395
(25) Sinha S, Lin G, Ferenczi K. The skin microbiome and the gut-skin axis. Clin Dermatol. 2021 39(5):829–839.
doi:10.1016/j.clindermatol.2021.08.021
(26) Salem I, Ramser A, Isham N, Ghannoum MA. The gut microbiome as a major regulator of the gut-skin
axis. Front Microbiol. July 2018 9:1459. doi:10.3389/fmicb.2018.01459
(27) Greene AK, Nelson AM. The gut microbiome in melanoma: A piece of a complex puzzle. JAMA
Dermatol. 2023 159(10):1044–1046. doi:10.1001/jamadermatol.2023.2952
(28) Maguire M, Maguire G. The role of microbiota, and probiotics and prebiotics in skin health. Arch
Dermatol Res. 2017 309(6):411–421. doi:10.1007/s00403-017-1750-3
(29) Codoñer FM, Ramírez-Bosca A, Climent E, et al. Gut microbial composition in patients with psoriasis.
Sci Rep. February 2018 8(1):3812. doi:10.1038/s41598-018-22125-y
(30) O’Neill CA, Monteleone G, McLaughlin JT, Paus R. The gut-skin axis in health and disease: A paradigm
with therapeutic implications. BioEssays. 2016 38(11):1167–1176. doi:10.1002/bies.201600008
(31) Stec A, Sikora M, Maciejewska M, et al. Bacterial metabolites: A link between gut microbiota and
dermatological diseases. Int J Mol Sci. February 2023 24(4). doi:10.3390/ijms24043494
(32) Tan J, McKenzie C, Potamitis M, et al. Insights into the gut-skin axis: implications for skin disorders.
Clin Microbiol Rev. 2018 31:e00101–18.
(33) Cummings JH, Macfarlane GT. Role of intestinal bacteria in nutrient metabolism. Clin Nutr.
1997 16:3–11.
(34) Chakraborty P, Banerjee D, Majumder P, Sarkar J. Gut microbiota nexus: exploring the interactions
with the brain, heart, lungs, and skin axes and their effects on health, Med. Microecol. March 2024 20.
(35) Kimura I, Ichimura A, Ohue-Kitano R, Igarashi M. Free fatty acid receptors in health and disease.
Physiol Rev. 2020 100(1):171–210. doi:10.1152/physrev.00041.2018
(36) Jiminez V, Yusuf N. Bacterial metabolites and inflammatory skin diseases. Metabolites. August 2023 13(8).
doi:10.3390/metabo13080952
(37) Huang Y, Liu L, Hao Z, et al. Potential roles of gut microbial tryptophan metabolites in the complex
pathogenesis of acne vulgaris. Front Microbiol. July 2022 13:942027. doi:10.3389/fmicb.2022.942027
(38) Roager HM, Licht TR. Microbial tryptophan catabolites in health and disease. Nat Commun. August
2018 9(1):3294. doi:10.1038/s41467-018-05470-4
(39) Jennis M, Cavanaugh CR, Leo GC, Mabus JR, Lenhard J, Hornby PJ. Microbiota-derived tryptophan
indoles increase after gastric bypass surgery and reduce intestinal permeability in vitro and in vivo.
Neurogastroenterol Motil. August 2018 30(2). doi:10.1111/nmo.13178
(40) Esser C, Rannug A. The aryl hydrocarbon receptor in barrier organ physiology, immunology, and
toxicology. Pharmacol Rev. 2015 67(2):259–279. doi:10.1124/pr.114.009001
(41) Yu J, Luo Y, Zhu Z, et al. A tryptophan metabolite of the skin microbiota attenuates inflammation
in patients with atopic dermatitis through the aryl hydrocarbon receptor. J Allergy Clin Immunol.
2019 143:2108–2119.e12.
(42) Liu X, Zhang X, Zhang J, et al. Activation of aryl hydrocarbon receptor in Langerhans cells by a microbial
metabolite of tryptophan negatively regulates skin inflammation. J Dermatol Sci. 2020 100(3):192–200.
doi:10.1016/j.jdermsci.2020.10.004
(43) Jux B, Kadow S, Luecke S, Rannug A, Krutmann J, Esser C. The aryl hydrocarbon receptor mediates
UVB radiation-induced skin tanning. J Invest Dermatol. 2011 131(1):203–210. doi:10.1038/jid.2010.269