545 Modern Skincare
years, phages have been investigated as an antibiotic alternative for a variety of different
applications across the globe, prompting careful consideration of how to expand access
to their use. This expansion has continued into the skin care industry, where there is a
desire to leverage a new appreciation of the skin microbiome into thoughtful microbiota
modulatory active ingredients.
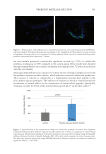
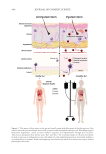
The studies described herein demonstrate the efficacy of this approach in mitigating acne
vulgaris via topical application of a cocktail of phage targeting C. acnes. The phage cocktail
performed exceptionally at the in vitro, ex vivo, and in vivo level with an excellent safety
profile. Examinations in the laboratory saw rapid clearance of bacteria in both planktonic
and biofilm conditions. Furthermore, the diminishment of the bacterial levels was rapid,
occurring within a 24 hour timeframe. In three-dimensional tissue modeling blemished
skin, the phage succeeded in reducing bacterial levels, a key inflammatory molecule, and once
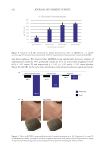
again showed no associated tissue toxicity. Finally, a limited pilot clinical study produced
evidence of improved skin conditions within 7 days, where sebum was reduced, the cognate
bacteria were diminished, and no significant fluctuation in neighboring bacterial groups
was observed. Like all the prior experiments, there was no reported adverse reactions. This
latter point has been emphasized as topically deploying bacteriophages (literally viruses that
consume bacteria) as a skincare microbiome modulatory ingredient is likely to be heavily
scrutinized for its safety profile. Their relative safety is unsurprising to microbiologists as
phages are the largest constituent of the planetary biosphere. Indeed, at any given time, there
are approximately 1031 phage particles on the planet, and as such, phages are quite literally
part of every environmental niche.64 And with the notion that phages are everywhere, they
are also naturally found on the skin. So, when phages are introduced to the skin as a bio-
active topical ingredient, it is not a foreign material to the tissue.
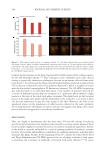
One particular aspect of the findings from these studies that deserves to be highlighted is the
impact that the phage cocktail had against C. acnes biofilms (Figure 4). Biofilms represent
the closest that prokaryotic microorganisms come to forming complex multicellular tissues
observed in higher eukaryotic organisms. Existing in a biofilm relative to a unicellular
planktonic state offers a variety advantages to such microbes, particularly a demonstrable
resistance to various antimicrobial measures.47 C. acnes has been established as a biofilm-
forming bacterial species, and its capacity to form these biological structures has been
suggested as a mechanism to circumvent topical antibiotics and as means to insulate the
anaerobic microbe from the oxygen rich environment outside the pilosebaceous unit where
it takes up residence in the skin.7,47–50 As such, it is highly fortunate that in the context of
multiple biofilm-forming pathogenic bacteria, phages (particularly a cocktail of multiple
distinct phages) have reproducibly demonstrated biofilm inhibition and disruption
efficacy.51–53 Consistent with those trends, the C. acnes phage cocktail utilized here showed
a dose-dependent reduction in C. acnes biofilms (Figure 4).
As the skin care industry considers utilizing this technological platform based on the
naturally occurring predators of bacteria, there are two different approaches that they can
embrace. The first involves the creation of a standardized cocktail of phages that has as broad
coverage as possible of the different strains of its cognate bacterial species—a “one-size fits
all” model. The alternative requires considerably greater investment in infrastructure and
time. This involves the development and delivery of “personalized” phage cocktails. For the
latter approach, an individual would submit a swab sample of the region of their skin for
which they would like to receive a phage-infused formulation. The sample would have the
strain diversity of the intended target bacteria in that skin region determined by genomic
years, phages have been investigated as an antibiotic alternative for a variety of different
applications across the globe, prompting careful consideration of how to expand access
to their use. This expansion has continued into the skin care industry, where there is a
desire to leverage a new appreciation of the skin microbiome into thoughtful microbiota
modulatory active ingredients.
The studies described herein demonstrate the efficacy of this approach in mitigating acne
vulgaris via topical application of a cocktail of phage targeting C. acnes. The phage cocktail
performed exceptionally at the in vitro, ex vivo, and in vivo level with an excellent safety
profile. Examinations in the laboratory saw rapid clearance of bacteria in both planktonic
and biofilm conditions. Furthermore, the diminishment of the bacterial levels was rapid,
occurring within a 24 hour timeframe. In three-dimensional tissue modeling blemished
skin, the phage succeeded in reducing bacterial levels, a key inflammatory molecule, and once
again showed no associated tissue toxicity. Finally, a limited pilot clinical study produced
evidence of improved skin conditions within 7 days, where sebum was reduced, the cognate
bacteria were diminished, and no significant fluctuation in neighboring bacterial groups
was observed. Like all the prior experiments, there was no reported adverse reactions. This
latter point has been emphasized as topically deploying bacteriophages (literally viruses that
consume bacteria) as a skincare microbiome modulatory ingredient is likely to be heavily
scrutinized for its safety profile. Their relative safety is unsurprising to microbiologists as
phages are the largest constituent of the planetary biosphere. Indeed, at any given time, there
are approximately 1031 phage particles on the planet, and as such, phages are quite literally
part of every environmental niche.64 And with the notion that phages are everywhere, they
are also naturally found on the skin. So, when phages are introduced to the skin as a bio-
active topical ingredient, it is not a foreign material to the tissue.
One particular aspect of the findings from these studies that deserves to be highlighted is the
impact that the phage cocktail had against C. acnes biofilms (Figure 4). Biofilms represent
the closest that prokaryotic microorganisms come to forming complex multicellular tissues
observed in higher eukaryotic organisms. Existing in a biofilm relative to a unicellular
planktonic state offers a variety advantages to such microbes, particularly a demonstrable
resistance to various antimicrobial measures.47 C. acnes has been established as a biofilm-
forming bacterial species, and its capacity to form these biological structures has been
suggested as a mechanism to circumvent topical antibiotics and as means to insulate the
anaerobic microbe from the oxygen rich environment outside the pilosebaceous unit where
it takes up residence in the skin.7,47–50 As such, it is highly fortunate that in the context of
multiple biofilm-forming pathogenic bacteria, phages (particularly a cocktail of multiple
distinct phages) have reproducibly demonstrated biofilm inhibition and disruption
efficacy.51–53 Consistent with those trends, the C. acnes phage cocktail utilized here showed
a dose-dependent reduction in C. acnes biofilms (Figure 4).
As the skin care industry considers utilizing this technological platform based on the
naturally occurring predators of bacteria, there are two different approaches that they can
embrace. The first involves the creation of a standardized cocktail of phages that has as broad
coverage as possible of the different strains of its cognate bacterial species—a “one-size fits
all” model. The alternative requires considerably greater investment in infrastructure and
time. This involves the development and delivery of “personalized” phage cocktails. For the
latter approach, an individual would submit a swab sample of the region of their skin for
which they would like to receive a phage-infused formulation. The sample would have the
strain diversity of the intended target bacteria in that skin region determined by genomic