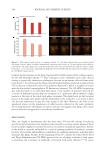
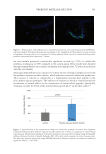
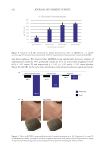
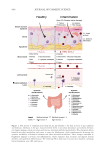
547 Modern Skincare
Both NHDF and HaCaT cell lines were cultured in Dulbecco’s Modified Eagle Medium
(DMEM, high glucose, pyruvate) with the addition of 10% fetal bovine serum (FBS),
10 mM N-2-hydroxyethylpiperazine-N-2-ethane sulfonic acid (HEPES), and 10 U/ml
Penicillin with 10 µg/ml Streptomycin (Pen/Strep). The nHEK cells were cultured in
EpiLife™ Medium, with 60 µM calcium and the addition of human keratinocyte growth
supplement. All growth mediums and their addition materials were purchased from
Thermo Fisher Scientific.
DOUBLE-LAYER AGAR (DLA) PLAQUE ASSAY
This assay allows for the calculation of the concentration, or titers, for a given phage sample
(Edvotek®, Washington, DC, USA). Five milliliters of the desired top agar was liquefied
and combined with 1 mL of bacterial culture and 100 µL of phage in a tube and gently
mixed without creating bubbles. The entire contents can then be poured over a LB agar
base plate. The plate is then gently but quickly tilted to ensure the entire base plate is
covered. Plates are allowed to solidify at room temperature. Once solidified, plates are
inverted and incubated overnight under anaerobic conditions at 37°C. After 24 hours,
check plates for any visible plaques. Plates should appear cloudy, with visibly cleared circles,
or plaques, throughout. Phage titers can be calculated by counting the number of visible
plaques, or PFU on a sample plate (PFU/100 µL), multiplying by any dilution factor that
was used on the phage sample itself, and then further multiplying by 1000 to convert from
microliters to milliliters. This calculated value provides the number of PFU/mL, otherwise
known as the phage titer.
CELL VIABILITY ASSAY
The relative cytotoxicity of the C. acnes phages on monolayers of skin cells was determined
using a resazurin viability assay. Briefly, cells were treated with varying concentrations of
individual phage material diluted in each designated growth media media without phage
was used as a control. Treated cells were incubated for 72 hours at 37°C with 5% CO
2 and humidity. Cell treatments were aspirated, and cells were washed twice with Hank’s
Balanced Salt Solution (HBSS, with calcium and magnesium, without phenol red) obtained
from Corning® (Corning, NY, USA). Wash HBSS was replaced with a 42 µM solution
of resazurin sodium salt in HBSS (resazurin sodium salt obtained from Sigma-Aldrich®
(St. Louis, MO, USA)) and placed at 37°C with 5% CO
2 and humidity, until a color change
was observed. After a color change was observed, samples of each solution were read on a
spectrophotometer with absorbance set to 570 nm and 690 nm. Optical density values at
690 nm were subtracted from 570 nm, and all test samples were compared to medium
control.
MINIMUM BIOFILM ERADICATION CONCENTRATION (MBEC) ASSAY
The triple phage cocktail was diluted from 4% to 0.03125% in bacterial growth media,
and the dilutions were tested against biofilms of C. acnes (#6919, ATCC) following the
protocol established by ASTM Standard E2799 (ASTM International, West Conshohocken,
Both NHDF and HaCaT cell lines were cultured in Dulbecco’s Modified Eagle Medium
(DMEM, high glucose, pyruvate) with the addition of 10% fetal bovine serum (FBS),
10 mM N-2-hydroxyethylpiperazine-N-2-ethane sulfonic acid (HEPES), and 10 U/ml
Penicillin with 10 µg/ml Streptomycin (Pen/Strep). The nHEK cells were cultured in
EpiLife™ Medium, with 60 µM calcium and the addition of human keratinocyte growth
supplement. All growth mediums and their addition materials were purchased from
Thermo Fisher Scientific.
DOUBLE-LAYER AGAR (DLA) PLAQUE ASSAY
This assay allows for the calculation of the concentration, or titers, for a given phage sample
(Edvotek®, Washington, DC, USA). Five milliliters of the desired top agar was liquefied
and combined with 1 mL of bacterial culture and 100 µL of phage in a tube and gently
mixed without creating bubbles. The entire contents can then be poured over a LB agar
base plate. The plate is then gently but quickly tilted to ensure the entire base plate is
covered. Plates are allowed to solidify at room temperature. Once solidified, plates are
inverted and incubated overnight under anaerobic conditions at 37°C. After 24 hours,
check plates for any visible plaques. Plates should appear cloudy, with visibly cleared circles,
or plaques, throughout. Phage titers can be calculated by counting the number of visible
plaques, or PFU on a sample plate (PFU/100 µL), multiplying by any dilution factor that
was used on the phage sample itself, and then further multiplying by 1000 to convert from
microliters to milliliters. This calculated value provides the number of PFU/mL, otherwise
known as the phage titer.
CELL VIABILITY ASSAY
The relative cytotoxicity of the C. acnes phages on monolayers of skin cells was determined
using a resazurin viability assay. Briefly, cells were treated with varying concentrations of
individual phage material diluted in each designated growth media media without phage
was used as a control. Treated cells were incubated for 72 hours at 37°C with 5% CO
2 and humidity. Cell treatments were aspirated, and cells were washed twice with Hank’s
Balanced Salt Solution (HBSS, with calcium and magnesium, without phenol red) obtained
from Corning® (Corning, NY, USA). Wash HBSS was replaced with a 42 µM solution
of resazurin sodium salt in HBSS (resazurin sodium salt obtained from Sigma-Aldrich®
(St. Louis, MO, USA)) and placed at 37°C with 5% CO
2 and humidity, until a color change
was observed. After a color change was observed, samples of each solution were read on a
spectrophotometer with absorbance set to 570 nm and 690 nm. Optical density values at
690 nm were subtracted from 570 nm, and all test samples were compared to medium
control.
MINIMUM BIOFILM ERADICATION CONCENTRATION (MBEC) ASSAY
The triple phage cocktail was diluted from 4% to 0.03125% in bacterial growth media,
and the dilutions were tested against biofilms of C. acnes (#6919, ATCC) following the
protocol established by ASTM Standard E2799 (ASTM International, West Conshohocken,