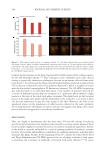
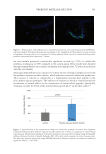
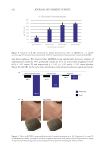
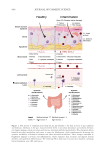
586 JOURNAL OF COSMETIC SCIENCE
built. The community includes the following species: Cutibacterium: C. acnes, C. avidum,
C. namnetense, C. Granulosum, Staphylococcus: S. epidermidis, S. capitis, S. aureus, S. Hominis,
Corynebacterium: C. tuberculostearicum, C. kroppenstedtii Micrococcus luteus Kocuria palustris
and rhizophila and Bacillus thuringiensis. Species were plated in BHI agar supplemented with
0.1% of Tween80 at 37°C in aerobic (Staphylococcus, Micrococcus, Corynebacterium, and Kocuria
species), or anaerobic conditions (Cutibacterium species). A single colony was picked from
each plate and grown in BHI, Thermo Fischer Scientific) liquid medium for all bacteria
except Corynebacterium species that were cultivated in a specific liquid medium (confidential
composition). Fifty µL of each culture was pooled in a single tube. The bacterial density of
each culture was assessed as colony forming units per volume unit (CFU/mL) after growth
in agar medium under the appropriate conditions (as described above). The pool of cultures
was subjected to two DNA extractions using Zymo BIOMICS™ DNA miniprep (Zymo
Research). These two DNA solutions from the same mock community were prepared for
sequencing using the same protocol as for the skin samples. Two amplification replicates
were performed for each DNA solution.
BIOINFORMATIC ANALYSIS
Samples were analyzed with the bioinformatics pipeline DADA2,18 which consists of a series
of steps to filter the raw sequences obtained through Illumina sequencing and a final step
to obtain the taxonomy of the sequences that have been filtered for microbial community
study. A metadata sheet was used to gather all information specific to samples, such as
participant characteristics (age, sex, skin phenotype), skin sites, and the corresponding
sequencing run. Sequences were filtered based on size, quality, and presence of primers.
The error rate was determined by gathering samples sequenced in the same run, and
the subsequent denoising was performed with the pseudo-pooling method. Taxonomic
assignment was achieved with the database Silva 138.1 (online resource for quality checked
and aligned ribosomal RNA sequence data) at the species level. If the algorithm did not
manage to provide a classification at the species level, the species annotation was built with
the corresponding genus combined to the mention spp (species plural, e.g. Staphylococcus
spp.). Amplicon sequence variants (ASVs) that were not assigned to any genera were excluded
from the analysis. ASVs classified as mitochondria and chloroplast at the family and class
level, respectively, were also discarded from the analysis. To make the data manipulation
easier and to exclude potential sequencing artifacts, ASVs having less than 10 reads in
the overall study or present in a single sample were also discarded. A cutoff of 5,000
reads was selected to avoid the loss of rare species. Rarefaction curves were generated to
ensure sufficient sequencing depth with the vegan package from the R program. The vegan
package provides tools for descriptive community ecology. It has the most basic functions
of diversity analysis, community ordination, and dissimilarity analysis.
NORMALIZATION OF THE SEQUENCING DATA
Sequencing data were treated according to four different workflows to check the impact of
normalization methods. Total Sum Scaling (TSS) was achieved with the function transform_
sample_counts and rarefied counts, with and without replacement, were achieved with the
rarefy_even_depth function. Both functions are from the phyloseq package. Phyloseq is
built. The community includes the following species: Cutibacterium: C. acnes, C. avidum,
C. namnetense, C. Granulosum, Staphylococcus: S. epidermidis, S. capitis, S. aureus, S. Hominis,
Corynebacterium: C. tuberculostearicum, C. kroppenstedtii Micrococcus luteus Kocuria palustris
and rhizophila and Bacillus thuringiensis. Species were plated in BHI agar supplemented with
0.1% of Tween80 at 37°C in aerobic (Staphylococcus, Micrococcus, Corynebacterium, and Kocuria
species), or anaerobic conditions (Cutibacterium species). A single colony was picked from
each plate and grown in BHI, Thermo Fischer Scientific) liquid medium for all bacteria
except Corynebacterium species that were cultivated in a specific liquid medium (confidential
composition). Fifty µL of each culture was pooled in a single tube. The bacterial density of
each culture was assessed as colony forming units per volume unit (CFU/mL) after growth
in agar medium under the appropriate conditions (as described above). The pool of cultures
was subjected to two DNA extractions using Zymo BIOMICS™ DNA miniprep (Zymo
Research). These two DNA solutions from the same mock community were prepared for
sequencing using the same protocol as for the skin samples. Two amplification replicates
were performed for each DNA solution.
BIOINFORMATIC ANALYSIS
Samples were analyzed with the bioinformatics pipeline DADA2,18 which consists of a series
of steps to filter the raw sequences obtained through Illumina sequencing and a final step
to obtain the taxonomy of the sequences that have been filtered for microbial community
study. A metadata sheet was used to gather all information specific to samples, such as
participant characteristics (age, sex, skin phenotype), skin sites, and the corresponding
sequencing run. Sequences were filtered based on size, quality, and presence of primers.
The error rate was determined by gathering samples sequenced in the same run, and
the subsequent denoising was performed with the pseudo-pooling method. Taxonomic
assignment was achieved with the database Silva 138.1 (online resource for quality checked
and aligned ribosomal RNA sequence data) at the species level. If the algorithm did not
manage to provide a classification at the species level, the species annotation was built with
the corresponding genus combined to the mention spp (species plural, e.g. Staphylococcus
spp.). Amplicon sequence variants (ASVs) that were not assigned to any genera were excluded
from the analysis. ASVs classified as mitochondria and chloroplast at the family and class
level, respectively, were also discarded from the analysis. To make the data manipulation
easier and to exclude potential sequencing artifacts, ASVs having less than 10 reads in
the overall study or present in a single sample were also discarded. A cutoff of 5,000
reads was selected to avoid the loss of rare species. Rarefaction curves were generated to
ensure sufficient sequencing depth with the vegan package from the R program. The vegan
package provides tools for descriptive community ecology. It has the most basic functions
of diversity analysis, community ordination, and dissimilarity analysis.
NORMALIZATION OF THE SEQUENCING DATA
Sequencing data were treated according to four different workflows to check the impact of
normalization methods. Total Sum Scaling (TSS) was achieved with the function transform_
sample_counts and rarefied counts, with and without replacement, were achieved with the
rarefy_even_depth function. Both functions are from the phyloseq package. Phyloseq is