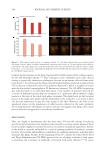
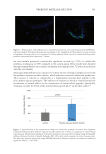
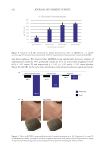
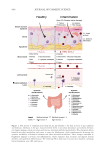
594 JOURNAL OF COSMETIC SCIENCE
genus and species levels to define a putative bacterial signature for SS, as this could help to
better understand and eventually improve this skin condition.
Unlike Keum’s study13 that showed no changes in Cutibacterium and Staphylococcus genus
levels, or Jarrin et al.16 who observed an increase in Cutibacterium and a nonsignificant
decrease in Staphylococcus, we demonstrated an increase in Cutibacterium and a decrease in
Staphylococcus genus abundance between cohorts. This could be explained by a difference
in cohort (location, size, and age classes) and analytical methods used for sequencing. Our
data more closely matched with Filaire et al.,20 who showed that in SS there was an increase
in Actinobacteria (phylum composed essentially by Cutibacterium and Corynebacterium
genera) and a decrease in Firmicutes (phylum composed among others by the Staphylococcus
genus). Although not significant in relative frequency (p 0.05), Cutibacterium, Paracoccus,
and Corynebacterium were also found increased by Bai et al.,15 whereas Staphylococcus were
found decreased. Interestingly, Zheng et al.14 also showed that Staphylococci, particularly
S. epidermidis, significantly decreased in SS (p =0.028). Moreover, as S. epidermidis decreased
gradually with the degree of skin sensitivity, they indicated a correlation between skin
microbiome changes and female LAST score ≥3.
To complete our analysis, we observed no change in abundance of Corynebacterium and a
decrease in Staphylococcus in SS subjects. This partially aligns with the findings of Bai’s
study,15 which indicated a trend towards an increase in Corynebacterium and a decrease in
Staphylococcus, or Jarrin’s study,16 who reported a significant increase in Corynebacterium, but
not significant decrease in Staphylococcus. As observed by Jarrin et al.,16 we also observed
an increase in Kocuria, Micrococcus, and Lactococcus genera (data not shown). However,
we also observed contrasting results compared to them for Cutibacterium, Bacillus, and
Lawsonella, whose abundance tend to increase in our SS subjects, whereas they indicated
that Lactobacillus was decreased (data not shown). This could be explained by the difference
in the cohort ages (as Jarrin et al. focused on 20–50 year old panelists, while we analyzed
18–77 year old panelists) and/or the analytical method (targeted V3-V4 16S rRNA variable
regions versus V1-V9).
Our results are also partly in accordance with Hillion et al.12 as we observed in SS an increase
in the abundance of Corynebacterium and Brevibacterium (data not shown) and a decrease in
Staphylococcus. Having a mixed cohort, we also observed an increase in Micrococcus that they
only observed in male SS.
Taken all together, these combined results seem to demonstrate a core group of genera
that were increased in SS including Corynebacterium, Kocuria, Micrococcus, Lactococcus, and
Brevibacterium, whereas Staphylococcus was decreased. Additional, large studies on other bacteria
such as Cutibacterium, Bacillus, or Acinetobacter would help to further refine their status in SS.
Going deeper to the strain level provides additional insight as it more precisely highlights
the changes within a genus. For Corynebacteria, we observed a 1.6-fold increase in
Corynebacterium kroppenstedtii, already the most abundant species in NS. Little is known
about the Corynebacteria genus members contribution in SS, however an increase in
Corynebacterium kroppenstedtii was previously correlated to skin redness showing that this
bacterium could be involved in both the redness and sensitivity of the skin.20 Ridaura et al.
also showed in mice with high-fat diets that certain Corynebacteria, such as Corynebacterium
accolens, could promote inflammation in an IL-23 dependent manner.21 Corynebacterium and
particularly Corynebacterium kroppenstedtii could thus be considered as a way for cosmetic
care products to address SS.
genus and species levels to define a putative bacterial signature for SS, as this could help to
better understand and eventually improve this skin condition.
Unlike Keum’s study13 that showed no changes in Cutibacterium and Staphylococcus genus
levels, or Jarrin et al.16 who observed an increase in Cutibacterium and a nonsignificant
decrease in Staphylococcus, we demonstrated an increase in Cutibacterium and a decrease in
Staphylococcus genus abundance between cohorts. This could be explained by a difference
in cohort (location, size, and age classes) and analytical methods used for sequencing. Our
data more closely matched with Filaire et al.,20 who showed that in SS there was an increase
in Actinobacteria (phylum composed essentially by Cutibacterium and Corynebacterium
genera) and a decrease in Firmicutes (phylum composed among others by the Staphylococcus
genus). Although not significant in relative frequency (p 0.05), Cutibacterium, Paracoccus,
and Corynebacterium were also found increased by Bai et al.,15 whereas Staphylococcus were
found decreased. Interestingly, Zheng et al.14 also showed that Staphylococci, particularly
S. epidermidis, significantly decreased in SS (p =0.028). Moreover, as S. epidermidis decreased
gradually with the degree of skin sensitivity, they indicated a correlation between skin
microbiome changes and female LAST score ≥3.
To complete our analysis, we observed no change in abundance of Corynebacterium and a
decrease in Staphylococcus in SS subjects. This partially aligns with the findings of Bai’s
study,15 which indicated a trend towards an increase in Corynebacterium and a decrease in
Staphylococcus, or Jarrin’s study,16 who reported a significant increase in Corynebacterium, but
not significant decrease in Staphylococcus. As observed by Jarrin et al.,16 we also observed
an increase in Kocuria, Micrococcus, and Lactococcus genera (data not shown). However,
we also observed contrasting results compared to them for Cutibacterium, Bacillus, and
Lawsonella, whose abundance tend to increase in our SS subjects, whereas they indicated
that Lactobacillus was decreased (data not shown). This could be explained by the difference
in the cohort ages (as Jarrin et al. focused on 20–50 year old panelists, while we analyzed
18–77 year old panelists) and/or the analytical method (targeted V3-V4 16S rRNA variable
regions versus V1-V9).
Our results are also partly in accordance with Hillion et al.12 as we observed in SS an increase
in the abundance of Corynebacterium and Brevibacterium (data not shown) and a decrease in
Staphylococcus. Having a mixed cohort, we also observed an increase in Micrococcus that they
only observed in male SS.
Taken all together, these combined results seem to demonstrate a core group of genera
that were increased in SS including Corynebacterium, Kocuria, Micrococcus, Lactococcus, and
Brevibacterium, whereas Staphylococcus was decreased. Additional, large studies on other bacteria
such as Cutibacterium, Bacillus, or Acinetobacter would help to further refine their status in SS.
Going deeper to the strain level provides additional insight as it more precisely highlights
the changes within a genus. For Corynebacteria, we observed a 1.6-fold increase in
Corynebacterium kroppenstedtii, already the most abundant species in NS. Little is known
about the Corynebacteria genus members contribution in SS, however an increase in
Corynebacterium kroppenstedtii was previously correlated to skin redness showing that this
bacterium could be involved in both the redness and sensitivity of the skin.20 Ridaura et al.
also showed in mice with high-fat diets that certain Corynebacteria, such as Corynebacterium
accolens, could promote inflammation in an IL-23 dependent manner.21 Corynebacterium and
particularly Corynebacterium kroppenstedtii could thus be considered as a way for cosmetic
care products to address SS.