623 Pentapeptide to Control Acne
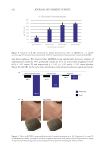
Clinical data are in-line with effect of pKTSKS in our in vitro experimentations and
demonstrate the efficacy of pKTSKS in acne-associated marks.
DISCUSSION
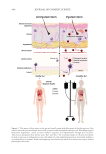
From the adolescence age, skin can present an unwanted and psychological challenging
aspect due to blemishes and scars resulting from the C. acnes proliferation in hair follicles
sheath. Sometimes protected into a biofilm, C. acnes strongly disturbs homeostasis of the hair
follicle sheath because of the bacterial cells production of extracellular lipases. These latter
create irritant molecules that result in the production of inflammatory mediators, inducing
keratinocyte activation, characterized by increased proliferation and a partial differentiation
process.28 Immature cells accumulate in the infundibulum and cause plugging. Release
of inflammatory mediators close to the hair follicle sheath induces expression of matrix
proteases and enzymes that target and degrade proteins of cutaneous tissues. Together they
cause blemishes such as inflammatory marks, pockmarks, and increase skin roughness.
The aim of this study was to find an acceptable and safe cosmetic active solution to prevent
the appearance of unpleasant acne lesions, but also to help skin to quickly recover its
smoothness by triggering synthesis of collagens and hyaluronic acid.
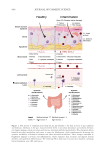
Because the increased quorum of bacteria9,10,29 is critical as it increases bacteria clustering
and biofilm production, we performed a screening on the capacity of different peptides to
inhibit C. acnes growth, inhibit production of proinflammatory mediators by keratinocytes
and induce production of extracellular matrix proteins by fibroblasts. We identified
pKTSKS, a lipo-pentapeptide, as the best candidate in these three parameters.
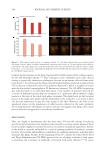
We showed that pKTSKS inhibits C. acnes growth both in vitro and in vivo. Isolated
compounds forming the lipo-pentapeptide (i.e. palmitic acid, KTSKS) are not efficient
on C. acnes growth, either alone or blended. pKTSKS did not act on S. epidermidis growth
as evidenced in a clinical test. Moreover, pKTSKS is more efficient on C. acnes ribotypes
RT-4 and RT-5 than on RT-1 strain, showing a strain-specific sensitivity to pKTSKS.
Explanations have yet to be found, and further studies need to be conducted to clarify this
observation. This lipo-pentapeptide did not have any remnant effect on C. acnes cells as
shown by lack of effect when lipo-pentapeptide was washed-out. This confirms the peptide
has no biocide activity and does not kill the cells.
We then focused on the effect of the pKTSKS on C. acnes biofilm and lipase. It was shown
that pKTSKS reduced adhesion of C. acnes on plastic support, one of the prerequisite steps
for its virulence and for biofilm production, suggesting an inhibition of quorum sensing.
Moreover, it was shown that this peptide reduced the activity of the C. acnes extracellular
lipases (data not shown) is another parameter of interest for reducing irritation and
downstream process leading to micro-inflammation of hair follicle sheath.13,14,29
Because persistence of inflammation is associated with both acne severity and scars, we then
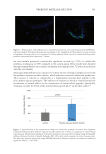
evaluated on anti-inflammatory effect of pKTSKS on different skin cells. This pentapeptide
reduces C. acnes-induced IL-6 release in both sebocytes and keratinocytes while it increases
the IL1-Ra release, limiting the proinflammatory cascade and its consequences. We also
tested the effect of pKTSKS in components of extracellular matrix components (ECM):
hyaluronic acid, collagen-I, collagen-IV, and fibronectin. Although hyaluronic acid is
synthesized in both the dermis and the epidermis, we opted to focus our investigation on
its role within keratinocytes due to its significant impact on hydration and skin barrier
Clinical data are in-line with effect of pKTSKS in our in vitro experimentations and
demonstrate the efficacy of pKTSKS in acne-associated marks.
DISCUSSION
From the adolescence age, skin can present an unwanted and psychological challenging
aspect due to blemishes and scars resulting from the C. acnes proliferation in hair follicles
sheath. Sometimes protected into a biofilm, C. acnes strongly disturbs homeostasis of the hair
follicle sheath because of the bacterial cells production of extracellular lipases. These latter
create irritant molecules that result in the production of inflammatory mediators, inducing
keratinocyte activation, characterized by increased proliferation and a partial differentiation
process.28 Immature cells accumulate in the infundibulum and cause plugging. Release
of inflammatory mediators close to the hair follicle sheath induces expression of matrix
proteases and enzymes that target and degrade proteins of cutaneous tissues. Together they
cause blemishes such as inflammatory marks, pockmarks, and increase skin roughness.
The aim of this study was to find an acceptable and safe cosmetic active solution to prevent
the appearance of unpleasant acne lesions, but also to help skin to quickly recover its
smoothness by triggering synthesis of collagens and hyaluronic acid.
Because the increased quorum of bacteria9,10,29 is critical as it increases bacteria clustering
and biofilm production, we performed a screening on the capacity of different peptides to
inhibit C. acnes growth, inhibit production of proinflammatory mediators by keratinocytes
and induce production of extracellular matrix proteins by fibroblasts. We identified
pKTSKS, a lipo-pentapeptide, as the best candidate in these three parameters.
We showed that pKTSKS inhibits C. acnes growth both in vitro and in vivo. Isolated
compounds forming the lipo-pentapeptide (i.e. palmitic acid, KTSKS) are not efficient
on C. acnes growth, either alone or blended. pKTSKS did not act on S. epidermidis growth
as evidenced in a clinical test. Moreover, pKTSKS is more efficient on C. acnes ribotypes
RT-4 and RT-5 than on RT-1 strain, showing a strain-specific sensitivity to pKTSKS.
Explanations have yet to be found, and further studies need to be conducted to clarify this
observation. This lipo-pentapeptide did not have any remnant effect on C. acnes cells as
shown by lack of effect when lipo-pentapeptide was washed-out. This confirms the peptide
has no biocide activity and does not kill the cells.
We then focused on the effect of the pKTSKS on C. acnes biofilm and lipase. It was shown
that pKTSKS reduced adhesion of C. acnes on plastic support, one of the prerequisite steps
for its virulence and for biofilm production, suggesting an inhibition of quorum sensing.
Moreover, it was shown that this peptide reduced the activity of the C. acnes extracellular
lipases (data not shown) is another parameter of interest for reducing irritation and
downstream process leading to micro-inflammation of hair follicle sheath.13,14,29
Because persistence of inflammation is associated with both acne severity and scars, we then
evaluated on anti-inflammatory effect of pKTSKS on different skin cells. This pentapeptide
reduces C. acnes-induced IL-6 release in both sebocytes and keratinocytes while it increases
the IL1-Ra release, limiting the proinflammatory cascade and its consequences. We also
tested the effect of pKTSKS in components of extracellular matrix components (ECM):
hyaluronic acid, collagen-I, collagen-IV, and fibronectin. Although hyaluronic acid is
synthesized in both the dermis and the epidermis, we opted to focus our investigation on
its role within keratinocytes due to its significant impact on hydration and skin barrier