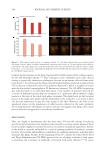
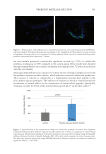
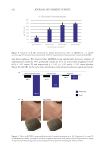
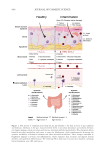
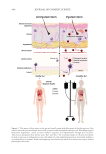
625 Pentapeptide to Control Acne
(13) Gannesen AV, Zdorovenko EL, Botchkova EA, et al. Composition of the biofilm matrix of Cutibacterium
acnes acneic strain RT5. Front Microbiol. 2019 10:1284. doi:10.3389/fmicb.2019.01284
(14) Miskin JE, Farrell AM, Cunliffe WJ, Holland KT. Propionibacterium acnes, a resident of lipid-
rich human skin, produces a 33 kDa extracellular lipase encoded by gehA. Microbiology (Reading).
1997 143(5):1745–1755. doi:10.1099/00221287-143-5-1745
(15) Rafferty J, Nagaraj H, McCloskey AP, et al. Peptide therapeutics and the pharmaceutical industry:
barriers encountered translating from the laboratory to patients. Curr Med Chem. 2016 23(37):4231–
4259. doi:10.2174/0929867323666160909155222
(16) Lintner K, Peschard O. Biologically active peptides: from a laboratory bench curiosity to a functional
skin care product. Int J Cosmet Sci. 2000 22(3):207–218. doi:10.1046/j.1467-2494.2000.00010.x
(17) Lintner K, Mondon P, Peschard O, Mas-Chamberlin C. Cosmetic applications of a wound healing
peptide. J Cosmet Sci. 2001 52:82–83.
(18) Mondon P, Fache S, Doridot E, Lintner K. From elastin to elastic fibers, Part II: The clinical effects of a
natural dipeptide on the biological cascade, Cosmet. Toilet Mag. 2012 127:658–664.
(19) Mondon P, Hillion M, Peschard O, et al. Evaluation of dermal extracellular matrix and epidermal-
dermal junction modifications using matrix-assisted laser desorption/ionization mass spectrometric
imaging, in vivo reflectance confocal microscopy, echography, and histology: effect of age and peptide
applications. J Cosmet Dermatol. 2015 14(2):152–160. doi:10.1111/jocd.12135
(20) Mondon P, Leroux R, Ringenbach C, Debacker A, Peschard O. A micropeptide modulating a-Crystallin,
ANCR and TRPC6 to improve skin barrier functions and homeostasis. IFSCC Mag. 2019 3:1–6.
(21) Fields GB, Noble RL. Solid phase peptide synthesis utilizing 9-fluorenylmethoxycarbonyl amino acids.
Int J Pept Protein Res. 1990 35(3):161–214. doi:10.1111/j.1399-3011.1990.tb00939.x
(22) Merritt JH, Kadouri DE, O’Toole GA. Growing and analyzing static biofilms. Curr Protoc Microbiol.
2005 Chapter(1):Unit 1B.1. doi:10.1002/9780471729259.mc01b01s00
(23) Boukamp P, Petrussevska RT, Breitkreutz D, Hornung J, Markham A, Fusenig NE. Normal
keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J Cell Biol.
1988 106(3):761–771. doi:10.1083/jcb.106.3.761
(24) Labarca C, Paigen K. A simple, rapid, and sensitive DNA assay procedure. Anal Biochem. 1980 102(2):344–
352. doi:10.1016/0003-2697(80)90165-7
(25) Asselineau D, Bernhard B, Bailly C, Darmon M. Epidermal morphogenesis and induction of the 67
kD keratin polypeptide by culture of human keratinocytes at the liquid-air interface. Exp Cell Res.
1985 159(2):536–539. doi:10.1016/s0014-4827(85)80027-6
(26) Christensen GJM, Scholz CFP, Enghild J, et al. Antagonism between Staphylococcus epidermidis
and Propionibacterium acnes and its genomic basis. BMC Genomics. 2016 17:152. doi:10.1186/
s12864-016-2489-5
(27) Moon J, Yoon JY, Yang JH, Kwon HH, Min S, Suh DH. Atrophic acne scar: a process from altered
metabolism of elastic fibres and collagen fibres based on transforming growth factor-β1 signalling. Br J
Dermatol. 2019 181(6):1226–1237. doi:10.1111/bjd.17851
(28) Isard O, Knol AC, Ariès MF, et al. Propionibacterium acnes activates the IGF-1/IGF-1R system in
the epidermis and induces keratinocyte proliferation. J Invest Dermatol. 2011 131(1):59–66. doi:10.1038/
jid.2010.281
(29) Mion S, Rémy B, Plener L, Chabrière É, Daudé D. Quorum sensing and quorum quenching: how to
disrupt bacterial communication to inhibit virulence? Med Sci (Paris). 2019 35(1):31–38. doi:10.1051/
medsci/2018310
(30) Pai VV, Bhandari P, Shukla P. Topical peptides as cosmeceuticals. Indian J Dermatol Venereol Leprol.
2017 February(1):9–18. doi:10.4103/0378-6323.186500
(31) Skibska A, Perlikowska R. Signal peptides – promising ingredients in cosmetics. Curr Protein Pept Sci.
2021 22(10):716–728. doi:10.2174/1389203722666210812121129
(13) Gannesen AV, Zdorovenko EL, Botchkova EA, et al. Composition of the biofilm matrix of Cutibacterium
acnes acneic strain RT5. Front Microbiol. 2019 10:1284. doi:10.3389/fmicb.2019.01284
(14) Miskin JE, Farrell AM, Cunliffe WJ, Holland KT. Propionibacterium acnes, a resident of lipid-
rich human skin, produces a 33 kDa extracellular lipase encoded by gehA. Microbiology (Reading).
1997 143(5):1745–1755. doi:10.1099/00221287-143-5-1745
(15) Rafferty J, Nagaraj H, McCloskey AP, et al. Peptide therapeutics and the pharmaceutical industry:
barriers encountered translating from the laboratory to patients. Curr Med Chem. 2016 23(37):4231–
4259. doi:10.2174/0929867323666160909155222
(16) Lintner K, Peschard O. Biologically active peptides: from a laboratory bench curiosity to a functional
skin care product. Int J Cosmet Sci. 2000 22(3):207–218. doi:10.1046/j.1467-2494.2000.00010.x
(17) Lintner K, Mondon P, Peschard O, Mas-Chamberlin C. Cosmetic applications of a wound healing
peptide. J Cosmet Sci. 2001 52:82–83.
(18) Mondon P, Fache S, Doridot E, Lintner K. From elastin to elastic fibers, Part II: The clinical effects of a
natural dipeptide on the biological cascade, Cosmet. Toilet Mag. 2012 127:658–664.
(19) Mondon P, Hillion M, Peschard O, et al. Evaluation of dermal extracellular matrix and epidermal-
dermal junction modifications using matrix-assisted laser desorption/ionization mass spectrometric
imaging, in vivo reflectance confocal microscopy, echography, and histology: effect of age and peptide
applications. J Cosmet Dermatol. 2015 14(2):152–160. doi:10.1111/jocd.12135
(20) Mondon P, Leroux R, Ringenbach C, Debacker A, Peschard O. A micropeptide modulating a-Crystallin,
ANCR and TRPC6 to improve skin barrier functions and homeostasis. IFSCC Mag. 2019 3:1–6.
(21) Fields GB, Noble RL. Solid phase peptide synthesis utilizing 9-fluorenylmethoxycarbonyl amino acids.
Int J Pept Protein Res. 1990 35(3):161–214. doi:10.1111/j.1399-3011.1990.tb00939.x
(22) Merritt JH, Kadouri DE, O’Toole GA. Growing and analyzing static biofilms. Curr Protoc Microbiol.
2005 Chapter(1):Unit 1B.1. doi:10.1002/9780471729259.mc01b01s00
(23) Boukamp P, Petrussevska RT, Breitkreutz D, Hornung J, Markham A, Fusenig NE. Normal
keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J Cell Biol.
1988 106(3):761–771. doi:10.1083/jcb.106.3.761
(24) Labarca C, Paigen K. A simple, rapid, and sensitive DNA assay procedure. Anal Biochem. 1980 102(2):344–
352. doi:10.1016/0003-2697(80)90165-7
(25) Asselineau D, Bernhard B, Bailly C, Darmon M. Epidermal morphogenesis and induction of the 67
kD keratin polypeptide by culture of human keratinocytes at the liquid-air interface. Exp Cell Res.
1985 159(2):536–539. doi:10.1016/s0014-4827(85)80027-6
(26) Christensen GJM, Scholz CFP, Enghild J, et al. Antagonism between Staphylococcus epidermidis
and Propionibacterium acnes and its genomic basis. BMC Genomics. 2016 17:152. doi:10.1186/
s12864-016-2489-5
(27) Moon J, Yoon JY, Yang JH, Kwon HH, Min S, Suh DH. Atrophic acne scar: a process from altered
metabolism of elastic fibres and collagen fibres based on transforming growth factor-β1 signalling. Br J
Dermatol. 2019 181(6):1226–1237. doi:10.1111/bjd.17851
(28) Isard O, Knol AC, Ariès MF, et al. Propionibacterium acnes activates the IGF-1/IGF-1R system in
the epidermis and induces keratinocyte proliferation. J Invest Dermatol. 2011 131(1):59–66. doi:10.1038/
jid.2010.281
(29) Mion S, Rémy B, Plener L, Chabrière É, Daudé D. Quorum sensing and quorum quenching: how to
disrupt bacterial communication to inhibit virulence? Med Sci (Paris). 2019 35(1):31–38. doi:10.1051/
medsci/2018310
(30) Pai VV, Bhandari P, Shukla P. Topical peptides as cosmeceuticals. Indian J Dermatol Venereol Leprol.
2017 February(1):9–18. doi:10.4103/0378-6323.186500
(31) Skibska A, Perlikowska R. Signal peptides – promising ingredients in cosmetics. Curr Protein Pept Sci.
2021 22(10):716–728. doi:10.2174/1389203722666210812121129