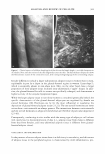
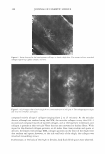
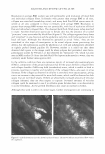
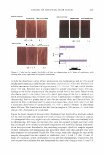
COSMECEUTICAL PROPERTIES OF LEVAN 397 N-(2-hydroxyethyl) piperazine-N' -(2-ethanesulfonic acid) (HEPES), and bovine serum albumin were from the Sigma Chemical Co. (St. Louis, MO). Dulbecco's Modified Eagle's Medium (DMEM), keratinocyte-serum free media (K-SFM), fetal bovine serum, and antibiotics were all purchased from Life Technologies, Inc. (Grand Island, NY). The insert used for the skin equivalent was acquired from Millipore (Bedford, MA). Type I collagen was purchased from Bioland (Bioland, Cheonan, Korea). The ELISA kit, which was used for the interleukin-la release assay, was purchased from Endogen, Inc. (Boston, MA). All of the remaining commercially available chemicals used in this study were reagent-grade, and were used as received. METHODS Preparation of levan from Zymomonas mobilis ( 19 J 20). Zymomonas mobilis ZMl (ATCC10988) was cultured using a 50-liter jar fermentor (KoBiotech Co., Inchon, Korea) with a 10-liter working volume at 30°C, at a pH of 5 .0, for 24 hours. The medium used in this study was composed of 10% sucrose, 1 % yeast extract, and 0.1 % KH2PO4 . After fermentation, the cells were removed from the culture medium, which contained 15 g/1 of levan, via centrifugation (3000 rpm, 10 min). The low-molecular weight by-products, including glucose, sucrose, and oligosaccharides, were then ex tracted via ultrafiltration (0.2-µm filter). The levan was precipitated by the addition of EtOH (EtOH: media = 3: 1 by weight). The levan precipitate was then resuspended in distilled water and settled out repeatedly, twice using the same method. The precipitate was finally dried at 80°C, yielding a powdered product. Analysis of levan structure. In order to characterize the structure of the levan, we recorded the 13C-NMR spectra on a Varian-Mercury Plus 400 spectrometer. The 13C-NMR spectra were then calibrated and reported, using TMS (tetramethylsilane) as an internal standard. The samples were dissolved in D 2 O (about 10 mg/0.5 ml of D 2 O). Our results were then compared with previously reported data (21,22). Molecular weight determination. Molecular weights were determined via gel permeation chromatography, using a Viscotek Tri-SEC system with a Viscotek T-60A dual detector (light scattering, viscometer), under the following conditions: a TOSOH GMPWXL column with a 0.2 M NaCl, 0.1 % NaN 3 aqueous solution eluent, at a 0.5 ml/min flow rate. Poly(ethylene oxide)s were used as standards for the molecular weights. Particle size measurement. The distribution of particle size was determined in water, using an ELS-8000 electrophoretic light-scattering spectrophotometer (Otsuka), a 632.8-nm He-Ne laser (10 mW), at 25°C, with a relative refractive index of 1.3313 (Marquardt analysis method, 100 repeated tests). Transmission electron microscopy. Negative-stain micrographs were prepared on copper grids that had been covered with carbon film. In order to prepare the samples, diluted drops of levan solution were pipetted onto carbon-coated copper grids, then allowed to dry slowly. The copper grids were then stained with 1 % uranyl acetate, and were viewed and photographed using a transmission electron microscope OEM 1010, JEOL, Japan) at an accelerating voltage of 80 kV. Stability test in ethanol. Aqueous levan solution, at a concentration of 5 % on a solid basis, was then mixed with graded concentrations of ethanol (ranging from 10 wt% to 50 wt% of EtOH, in water). Turbidity and precipitation characteristics were observed in order to determine storage stability at room temperature, at 5 °C, and at 40°C, for a total of 45 days.
398 JOURNAL OF COSMETIC SCIENCE Measurement of moisturizing effect. Transepidermal water loss and water content were then assessed in order to evaluate the moisturizing effects of levan: (a) Measurement of transepidermal water loss (TEWL) using a Vapometer (23). A group of female volunteers, ranging in age from 22 to 3 7 years, were recruited into this study. Each of these volunteers was then familiarized with the transepidermal water loss (TEWL) method, so as to reduce any emotional stress that might be associated with the testing procedure. Additionally, the volunteers were requested to shave 24 hours prior to the test, and to avoid the intake of any food or drink that contained high levels of stimulatory caffeine. In an attempt to preclude any artificial effects on TEWL resultant from sweating, the volunteers were all instructed to apply an antiperspirant product prior to testing. The TEWL measurements were then conducted in a climate-controlled room, at a temperature between 20° and 25°C, with a relative humidity of 50±5%. Each of the volunteers spent 20 minutes of the equilibration period in the room, relaxing in a prone position with their hands behind their heads, exposing their underarms to the air. TEWL was measured with a Vapometer (Delfin Technologies Ltd.). The TEWL measurements were sequentially recorded at the application sites prior to treatment. These initial measurements were used as pre-treatment control values. Sub sequently, 20 µl of the sample was applied over a 4-cm2 area of the volar forearm (2 x 2 cm), followed by measurements taken at regular intervals for a total of six hours. (b) Measurement of skin moisture content using a Corneometer CM825. A skin hydra tion reading of each sample was recorded with a Corneometer CM825 (Courage Khazaka, West Germany). This equipment consisted of a recording device and an impedance probe that measures electrical conductivity on the surface of the skin. Capacitance refers to the quantity of electric changes stored, and thus capacitance is proportional to the amount of water in the skin, a factor that commonly referred to as skin hydration. Simply put, the higher the level of skin moisture, the stronger the observed conductance signal will be (24). Baseline values were taken from ten female volunteers aged between 22 and 3 7 years, using 40-mm-diameter circular test areas on both forearms. These panelists remained at rest in a room at a temperature of 25°C, with 45-55% relative humidity, for the duration of the test. Then, each of the designated areas was treated with a 10-µl/circle of five different test formulations. Cytotoxicity assay. Human fibroblasts and keratinocytes were utilized in our assessment of the cytotoxicity of levan (5 % (w/w)). Each of the cell lines was inoculated on a 96-well plate supplemented with 100 µl of DMEM (Dulbecco's Modified Eagle Medium) con taining 10% FBS (fetal bovine serum, GIBCO BRL), with a density of 104 cells per well. These plates were then incubated for 24 hours at 3 7 ° C in an atmosphere containing 5 % CO 2 . After the addition of the levan solution, the cells were incubated for another 24 hours. The viability and proliferation of the cells were then measured via MTT assay. The MTT assay allows for convenient assays using MTT (3-(4,5-dimethylthiazol-2-yl)- 2,5-diphenyl tetrazolium bromide, yellow), which results in the generation of a water insoluble formazan dye (purple) upon bioreduction in the presence of mitochondrial dehydrogenase in viable cells (25 ). MTT solution (100 µl) was added to each well, then incubated for four hours. One hundred milliliters of 0.0lM HCI containing 10% SDS was also added. The quantity of formazan in the culture medium was determined
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)