J. Cosmet. Sci., 56, 407-425 (November/December 2005) Fading of artificial hair color and its prevention by photofilters B. LOCKE and J. JACHOWICZ, International Specialty Products, Wayne, NJ 07470. Accepted for publication August 9, 2005. Synopsis Fading of artificial hair color has been investigated by simulating actual usage conditions through exposure to artificial radiation in a weatherometer, with 0.35 mW/(m2 nm) at 340 nm, for 16 to 48 hours, and by periodical washing. Hair color was produced by using commercial two-part, permanent hair dyes with light auburn, medium auburn, and dark auburn shades. Formulations based on red couplers, such as 4-amino- 2-hydroxytoluene and 1-naphtol, as well as primary intermediates, such as l-hydroxyethyl-4,5-diamino pyrazole sulfate, were employed. Results indicate that the extent of fading, as measured by the total color change parameter, dE, is greatest for colored hair subjected to both irradiation and shampooing, and significantly smaller for hair undergoing only irradiation or washing. Color loss has been also found to be dependent upon the hair type employed, with colored natural white and bleached hair undergoing much greater change than colored brown hair. It has been also shown that hair color based on pyrazole interme diates displayed the deepest fading as a result of shampooing (dE-4-6 after ten shampooings) and irradia tion/shampooing (dE-14-16 after 32 hours of light exposure and four shampooings). The contribution of UV light (UVB + UV A) to the artificial hair-color loss was found experimentally to be dependent upon the irradiation dose and varied from 63% at 16 hours of irradiation time to 27% at 48 hours of light exposure. The theoretical extent of photoprotection by a formulation was assessed by calculating the percentage of UV light it attenuates in the wavelength range from 290 nm to 400 nm. The results indicate that UVB photofilters, such as octyl methoxy cinnamate, absorb less than 25% of the total UV irradiation at concentrations as high as 30 mg/(g hair). UVA absorbers were found to be more effective, with benzo phenone-3 and benzophenone-4 absorbing about 40% of UV at the same concentration. Corresponding experimental data were in reasonable agreement with the theoretical predictions. The data are also presented for color protection with treatments containing two photo-absorbers: benzo phenone-3-ZnO benzophenone-4-ZnO octyl methoxy cinnamate-ZnO and dimethylpabaimidopropyl laurdimoni um tosy late-benzophenone-3. INTRODUCTION Hair photodamage was shown to consist of a number of concurrent processes that result in chemical and physical changes in fiber properties (Figure 1). Lipid oxidation, disulfide bond cleavage, tryptophan degradation, and cysteic acid formation lead to an increase in fiber porosity, loss of mechanical strength, and an increase in surface roughness (1-3). The most perceptible is the change in fiber coloration as a result of photoreactions of the natural pigments, either eu- or pheomelanin (4,6), as well as photodecomposition of 407
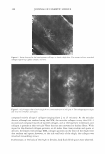
408 JOURNAL OF COSMETIC SCIENCE •Lipid Oxidation (increase in surface roughness) •Trp and Kynurenine Degradation •Disulfide Bond Cleavage •Cysteic Acid Formation •Artificial Haircolor Fading 1 Fiber Surface Damage I I 100 15000 200 •Decrease in Fiber Mechanical Strength •Natural Color Fading •Secondary Grooming Damage l Damage of Fiber Interior 300 400 (hrs) 600000 (J/cm1) Time/Dose (hrs )/(J/cm2) Figure 1. Hair structural photodamage and color fading as a function of irradiation dose. artificial hair dyes (6-9). As far as the latter is concerned, we refer to permanent oxidative hair color, which is expected to last indefinitely and resist washing and weathering. Other types of hair color, such as semipermanent and temporary treatments, are characterized by limited durability. Semi-permanent hair color can be removed by eight to ten shampooings, while temporary coloration is typically washed off in a single shampooing. The time scales of photoreactions of melanins and synthetic oxidative hair dyes are different. Both eu- and pheomelanins require exposure times on the order of at least two hundred hours to produce a perceptible shift in color shade, especially for highly pigmented fibers. In addition, natural pigmentation is resistant to washings and does not fade even after multiple applications of these processes. In contrast to this, artificial oxidative hair color fades at a much faster rate, with dark auburn shades typically undergoing perceptible lightening within 8-10 hours of irradiation and one shampoo application. It is also worth mentioning that that it has been found that artificial coloration has a photoprotective effect on hair keratin (10, 11). It is generally agreed that artificial hair color instability, caused by weathering and habitual grooming practices, has several underlying components, such as (a) removal of a dye from hair during shampooing, (6) photodecomposition of hair color chromophores as a result of irradiation, and (c) thermal decomposition of the dye in the dark. In addition to this, the hair-coloring dyes exhibit different sensitivities to the UVB (280 nm-320 nm), UVA (320 nm--400 nm), visible (370 nm-780 nm), and IR (750 nm- 2800 nm) portions of solar radiation (Figure 2). It has been demonstrated in the literature that visible and UV A light are mostly responsible for the photofading of artificial hair color (6).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)