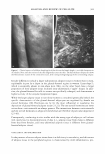
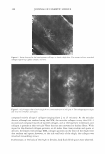
432 JOURNAL OF COSMETIC SCIENCE To qualify for the study, the backs of both hands of each participant had to have a visual scaling score of 2::1.5 and ::S2.5. On the backs of both hands of each panelist, two 2.5-cm-diameter circles were marked. The baseline condition of each site was then evaluated using a conductance meter (Skicon® 200 with an MT probe) and a trained observer. Each subject was then given Dial® liquid soap and hand-washing instructions. At the testing laboratory, subjects washed each hand with Dial® liquid soap at intervals of approximately one hour, five times each day for four days. The hand-washing instructions required subjects to wash each hand, individually, for 60 seconds, and then both hands were rinsed for 15 seconds with warm water (� 38° C). Following this, the backs of the hands were air-dried. After each of the first four ( 4) times the hands were washed and dried, a technician applied 10 µl (or approximately 1 mg/cm2) of test lotion to its assigned area. Assignment of three test products (lotions GR, W, and H) was based on a balanced complete-block design. The fourth site was left untreated ("no product" control). The fifth washing each day was used to eliminate residual test lotion that could hinder assessment. One hour after the fifth wash, during which panelists had re-acclimated in a controlled environ ment for at least 15 minutes, a clinical grader visually evaluated the hands for observable scaling, and Skicon® measurements were retaken. In doing the evaluations, the experienced evaluator terminated any panelist from the study who received a scaling score of 4 at any of the test sites. At this time, a D Squames® patch (used for the sampling of scaly skin) was collected for staining. The terminal measurements for each time were carried forward throughout the balance of the study. No additional baseline evaluations were carried out after Day 0. On Day 3, after the last hand-washing and drying procedure, D-Squames® tapes were collected for staining and color evaluation (squamometry). These D-Squames® tapes were evaluated using color imeter values (C*). Statistics. The test lotions' ability to reduce or reverse the onset of skin dryness and dehydration (evaluated by conductance) was assessed as a "change from the baseline" scores/measurements. Comparisons of this data to that of the "no product" control site represent the effects of the test lotion on the skin. The changes from baseline for the "no product" control reflect the effect of repeated hand washing on the skin. For daily skin hydration, as measured by Skicon® conductance, a two-way repeated measure ANOV A was used for analysis. The significance level was set at p ::S 0.05. Similarly, Skicon® conductance measurements of all termination scores were analyzed using a one-way, within-subject, ANOV A. The significance level was also set at p ::S 0.05. For each set of measurements, if an overall significant difference was observed (p ::S 0.05), the test products and control site were compared using the Tukey HSD test. For observer scoring of dryness, a similar statistical approach was used. The ability of the lotions to reduce the degree of damage to surface corneocytes was measured using stained D-Squames®. Day 3 results were compared with each other and with the untreated areas using a one-way, within-subject, ANOVA. The significance level was set at p ::S 0.05. When an overall significant difference was observed (p ::S 0.05 ), the Tukey HSD test was used to compare the test products and control site.
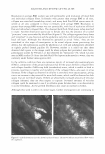
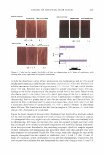
MOISTURIZER EFFICACY 433 MODEL SYSTEM III. BENEFIT: PREVENTION OF PRIMARY IRRITATION SURFACTANT PATCH TEST Test history. Primary skin irritation can be induced by many means. These include short-term (acute) exposure to a strong irritant or repeated (chronic) exposures to a weaker, cumulative irritant. Both approaches have been used experimentally. Kligman and Wooding (15), Phillips et al. (16), Frosch and Kligman (10), and Simian et al. (17) all describe patching methods that can produce primary irritation. Justice et al. (18) and Paye et al. (]_ 9) describe an arm-immersion test that produces erythema. As the purpose of the study was to assess the ability of moisturizers to prevent primary irritation, occlusive patching with sodium lauryl sulfate (SLS) was used to induce the irritation. Pretreatment of the skin with lotion was used to assess the ability of that product to prevent irritation, when compared with skin that was not pretreated. Similar methods have been used to assess the ability of barrier creams to reduce or even prevent the induction of irritation (20). For instance, Schentz et al. (21) standardized the repeated short-term occlusive irritation test (ROIT) to assess the ability of test-barrier creams to reduce irritation induced by toluene or 0.5% aqueous solutions of SLS. Sites on the volar forearm were pretreated with the test creams at 25 mg/cm2 . Ten minutes later the irritants were applied under occlusion for 30 minutes. This was repeated 3.5 hours later, and the twice-a-day treatment was used for two weeks (excluding the weekend). All four test centers found that the petrolatum-based creams were effective at reducing SLS-induced irritation as measured by TEWL rates and erythema measure ments (by both trained observer and colorimeter), but appeared to have a less consistent protective effect against toluene, possibly due to the hydrophobic nature of both pet rolatum and the solvent. Wigger-Alberti et al. (22) used a similar approach. They applied petrolatum to sites on the ventral forearms of 20 volunteers. Thirty minutes later, test irritants such as aqueous solutions of lauryl sulfate, sodium hydroxide, and lactic acid, as well as toluene, were applied for 30 minutes. This process was repeated daily for two weeks. Assessments of skin condition by erythema scoring, chromameter, and TEWL demonstrated that petrolatum could reduce irritation, although it appeared least effective against lactic acid. Procedure. Panelists whose skin was easily irritated by surfactants were recruited for this study. After they gave informed consent, three test sites, each 2 x 3 cm, were marked on each volar forearm. The test sites were evaluated for erythema, using a 0-to-4 scale (see below), and then two test products (lotions GR and H) were applied to their designated sites, the order being reversed on contralateral arms. The middle test site on each arm was a "no product" control. Each product was applied at a dosage of 2 mg/cm2 . After the test products had dried for at least 30 minutes, a 0.5% aqueous SLS solution was applied to each test site, using an occlusive 25-mm Hill Top® chamber system (Table III). After 24 hours, the Hill Top® chambers were removed and the test sites were rinsed with warm tap water and patted dry. A trained observer assessed erythema at each test site 24 hours after the patches were removed, using the same scale as shown above. Statistics. The erythema responses for two sites for each product were averaged and then compared to the responses for the other product and the untreated site using a paired t-test, with the p value set at 0.013 to compensate for multiple comparisons (Bonferroni).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)