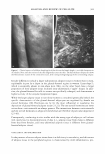
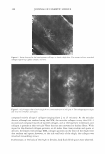
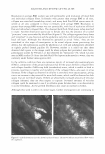
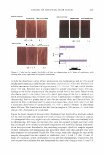
434 JOURNAL OF COSMETIC SCIENCE Erythema score* 0 2 3 4 * Half scores permitted. Table III Erythema Scoring Scale Description of skin None Minimal, non-uniform Moderate, uniform Severe Fiery red MODEL SYSTEM IV. BENEFIT: REDUCTION IN PRE-EXISTING PRIMARY IRRITATION 5%IVORY® SOAPSTUDY Test history. Loden and Anderson (3) investigated whether lipids used in moisturizers affect the skin's recovery after irritation with SLS. In their study, Loden and Andersson topically applied a variety of lipids to the SLS-irritated skin of subjects. Using 1 % hydrocortisone as the effective control, some lipids such as phytosterols were able to alleviate erythema. Our study protocol incorporated a modification of their methodol ogy, substituting 5% Ivory® soap for 0.5% SLS to give a less intense, more consistent erythema reaction. Panelists whose skin was easily irritated by surfactants were recruited for this study. Trained observers, using a 0-to-4 scale (shown above), rated the skin after it had been acclimated for at least 30 minutes ( 71 °F, RH = 3 5 ± 10% ). To be included in the study, panelists had to be erythema-free (erythema score = 0) at each of eight test sites (four per volar forearm). Twenty-five qualified panelists participated in the study. Each of eight test sites was patched with 5% Ivory® soap solution using an occlusive, 25-mm Hill Top® system. Patches remained in place for 22 hours. After the patches were removed, the test sites were rinsed with warm tap water, and then patted dry. Ninety minutes later, the panelists returned to the test laboratory, where they re-acclimated for at least 30 minutes under the environmental conditions described above, before their skin was re-evaluated for erythema. Test sites that showed sufficient irritation (erythema 2::: 1.0 on a 0-to-4 scale) were then treated with an application of 100 ul of test material. The test sites were protected by non-occlusive 25-mm Hill Top® chambers, each of which had a ¼-inch hole punched in the top, and from which the webril patches had been removed. After 22 hours, the protective chambers were removed and the test sites were rinsed and patted dry. Ninety minutes later, the panelists returned to the test laboratory, where they re-acclimated for at least 30 minutes under the environmental conditions described above. Finally, the test sites were re-evaluated for erythema by a trained observer. Statistics. Erythema reduction was determined from the difference in erythema before and after the test moisturizer was applied to the irritated skin (i.e., erythema score on Day 2 minus that on Day 3 ). Study validation required a significantly greater reduction in erythema by 1 % hydrocortisone (maximum level permitted by the FDA's over-the-
MOISTURIZER EFFICACY 435 counter regulations) compared with water. Significance was determined using a paired t-test. The ability of the test products to reduce erythema was compared using a one-way, within-subject, ANOVA. If an overall difference were detected (p � 0.05), then the individual means were compared using a Tukey HSD test. VALIDATION HOME-USE STUDY Study history. Ultimately, the clinical models are only useful if they predict the effects observed in real life. Therefore, we needed to assess whether the prediction that the high-glycerin lotion (GR) was more effective at reducing skin dryness and erythema than a lotion with much lower levels of glycerin is observed in a home-use situation. Han nuksela and Kinnunen's investigation (4) of whether moisturizers could prevent irritant contact dermatitis (ICD) caused by a detergent was used as a basis for our home-use study. Also, Zhai et al. 's studies (5 ,8), which reported on the use of dimethicone containing skin lotions to reduce SLS-induced irritant contact dermatitis (ICD) were again utilized in this study' s design. Procedure. Sixty volunteers, both male and female, ranging in age from 18 to 90 years, participated in a two-week, double-blind, between-groups, home-use study. They were in good general health and had no skin diseases other than mild eczema on the target site, and experienced itching with minimal cutaneous findings. Panelists were not pregnant, breast feeding, or undergoing treatment for any skin condition on the body did not have a history of allergic responses to sun exposure did not have a history of photosensitive skin conditions were not allergic to any ingredient in the products and were not taking systemic analgesic or antihistamine prescriptions. They had to be willing (a) to refrain from using unapproved topical nonprescription products and pre scription products on the body, and to use only the study products (6) to sign an informed consent statement and photography release form (c) to go to the research facility on assigned days at assigned times until the study was completed and (d) to refrain, throughout the study, from intentional sun exposure and the use of sun lamps and tanning beds, and the use self-tanning creams and other substances designed to artificially pigment the skin. They could not concurrently participate in another research study. Participants were equally split into two groups. One group used lotion GR and its associated mild liquid body and facial cleansers, while the other group used lotion LG, which has a substantially lower level of glycerin, and the leading U.S. liquid body and facial cleansers. All panelists were given one of two over-the-counter (OTC) analgesics for equal use on contralateral sites, as needed. At the onset and at the end of the study, photographs were taken of the target sites on ten subjects. Subjects did not apply lotions or creams for 12 hours before the first application of the study products. Additionally, except for the face and hands, the body was not washed, and no therapeutic or ancillary products were used for two hours before the first application of the study products. Upon their arrival at the test laboratory, panelists completed an informed consent form, and then acclimated their skin ( 21 °C at 30 ± 5 % RH) in an environmentally controlled room
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)