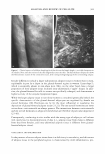
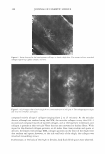
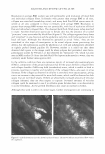
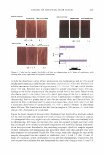
472 JOURNAL OF COSMETIC SCIENCE THE NOSE KNOWS OR WORKING AT THE "OLD FACTORY" FORMULATION PARAMETERS FOR NASAL DELIVERY SYSTEMS Gary Agisim, Richard Kenny, Sara Magee and Bhalchandra Patel Wyeth Consumer Healthcare Cosmetic chemists, in their role as formulation scientists, are increasingly asked to develop drug delivery systems and products for specialized anatomical sites, in particular the nose. In addition to its important olfaction sensory function, the nose is a route of increased interest for drug delivery, both via the nasal capillary system into the blood stream, and directly into the brain via the olfactory nerve which is not a true cranial nerve but a brain appendage. The nose is also a prominently visible organ requiring specialized care during cough/cold season. The design of nasal delivery systems must consider relevant physiological factors such as nasal anatomy especially site of drug deposition, blood flow, ciliary clearance, enzymatic activity, and pathophysiology of the nose. The key formulation properties include drug concentration,, dose, spray volume, droplet size/distribution, pH buffering, osmolarity, solubilization strategies, taste/throat sensation, preservation, humectants, viscosity, absorption promotion, and packaging components/materials, including labeling stock. Nasal drug delivery provides a viable alternative for the administration of many pharmaceutical agents. Some of the major advantages offered by the nasal route are: [l] --- Rapid absorption, higher bioavailab_ility, hence lower doses --- Fast onset of therapeutic action ---Avoidance of liver fust pass metabolism ---Avoidance of catabolism & irritation by gastrointestinal tract --- Minimally invasive, hence reduced risk of infection --- Convenient for self-medication improved patient compliance ANATOMY The nasal cavity is subdivided along the center into two halves by the nasal septum. The two cavities open to the facial side through the anterior nasal apertures and to the rhinopharaynx. via the posterior nasal apertures. The total surface area of the nasal cavity in man is about 150 cm2 and the total volume about 15 ml. Each of the two nasal cavities can be subdivided into three regions: the nasal vestibule, the olfactory region, and the respiratory region. The olfactory region in man covers and area of about 10 cm2 and is positioned on the superior turbinate, opposite the septum. The respiratory region is dominated by the large inferior turbinate. In the respiratory region, which is considered the major site of drug absorption into the systemic circulation, the mucosa consists of an epithelium resting on a basement membrane and a lamina propria. The anterior part of the respiratory regions is covered with squamous epithelium which changes to a transitional epithelium and converts in the posterior part of the cavity to pseudostratified columnar epithelium. The pseudostratified epithelium, also named respiratory epithelium, consists of four dominant cell types: ciliated columnar cells, non-ciliated columnar cells, goblet cells and basal cells. A total of 15 to 20% of the respiratory cells are covered by a layer of long cilia 2 to 4µm in size. The cilia move in a coordinated way to propel mucus across the epithelial surface to the pharynx. The respiratory cells are also covered by about 300 microvilli per cell. Microvilli increase the surface area of the cell considerably, which in tum promotes the transport of substances and water between the cells. Goblet cells are interspersed between the columnar cells and are the main entities responsible for the secretion of the mucus covering the epithelial cell layer. The mucus layer consists of a low viscosity sol layer that surrounds the cilia and a more viscous gel layer forming a layer on top of the sol layer and covering the tips of the cilia. The epithelial cells are closely connected on the apical surface, surrounded by intercellular junctions whose specialized sites and structural components are known as the junctional complex. Each complex is composed of three regions: Zonola Occludens, closest to the apical surface further down the zonola adherens and last the macula adherens. The zonola occludens forms a tight band around the upper part of the cell and contains the ZO-1 protein that influences diffusion of ions and neutral molecules through intercellular spaces. [2]
2005 ANNUAL SCIENTIFIC SEMINAR The mucus is a drixotropic, pseudoplastic sol/gel composed of 90-95% water, 1-2% salt, 2-35 mucin. The mucus has eight key functions: [3] --- Protects 1D1JCosa --- Retainer for substances in nasal duct -Adhesive --- Holds water --- Transports particulates --- Provides for transfer of heat --- Acts as a penneable matrix mesh --- Exhibits surface electrical activity FORMULATION CONSIDERATIONS Formulation pH and Buffering The formulation pH should be optimized: --- To avoidlminimize irritation of the nasal mucosa To achieve efficient drug absorption --- To promote optimal functioning of'the preservation system In general, to avoid nasal initation, the formulation pH should be adjusted to 4.5 to 6.5. The nasal surface pH is 7.39, and the pH of the nasal secretions is 5.5 -6.5 in adults and 5.0- 6.7 in infants. [ I] Most water-soluble drugs are absorbed in the unionized form. The most important factors in drug absorption and membrane transport are pH, pK, and drug partition coefficients. Most water soluble drugs are absorbed well in their unionized form. Hirari [4] found that nasal absorption of weak electrolytes such as salicylic acid and aminopyrine are highly dependent on the degree of ionization as predicted by Henderson-Hasselbach equation. For the slightly basic drug, arninopyrine, the absorption rates increase with increasing pH, suggesting that aminopyrine is absorbed through the nasal mucosa via passive transport of the unionized species. For salicylic acid the absorption increases with decreasing pH, as expected however, the experimental results indicate a substantially higher absorption than would be predicted by the pH/partition coefficient-lipoidal-transport model, indicating that perhaps salicylic acid promotes its oWn absorption through membrane disruption. Buffer systems used most frequently are sodium phosphate (dibasic)/ sodium phosphate (monobasic), sodium citrate/ citric acid, sodium borate/boric acid, glycine, sodium bicarbonate. Citrate offers the most optimal pKa (6.40) and synergistic antioxidant properties however, citrate salts can "sting" mucosa and are not fully compatible with benzalkonium chloride the most widely used preservative. Glycine is the least irritating, but with pK.'s of2.35 and 9.78 offers no buffering at the target 4.5 to 6.5 range. Borate has a pK. of9.24 and reacts with glycerin bicarbonate is unstable with the pH eventually drifting to 8.0 as the carbon dioxide volatilizes. Phosphate is the best compromise between buffer efficacy and irritation potential. Phosphate systems have a pK. of 7 .21. and are intermediate in irritation between citrate and glycine. A buffer capacity of 0.015 is very satisfactory and will maintain the pH of the formulation throughout shelf life. Most nasal sprays are formulated to isotonicity (285 mOsm/kg) to minimize irritation potential. Hypertonic solutions, up to 600 mOsm/kg, have been marketed with reports of increased nasal secretions, Although sodium chloride is the most widely used tonicity adjuster, reduced irritation has been reported with glycols particularly glycerin and sorbitol. [3] Preservation Benzalkonium Chloride is the preferred, and most frequently used, preservative in nasal sprays at a level 0.01 to 0.03%. This preservative is active against a wide range of bacteria, yeasts and fungi. Greater activity is seen with Gram-negative than with Gram-positive bacteria, with minimal activity against 473
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)