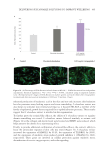
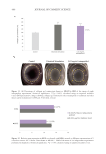
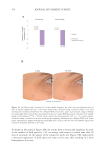
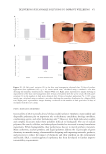
453 DELIVERING SUSTAINABLE SOLUTIONS TO IMPROVE WELLBEING
fluorescence intensity is proportional to the amount of the product in the PCR reaction.
Cycling conditions in the Bio-Rad CFX96 instrument are as follows: 95°C for 3 minutes,
followed by 40 cycles of denaturing at 95°C for 5 seconds, and annealing and elongation at
60°C for 30 seconds. GAPDH (Glyceraldehyde 3-phosphate dehydrogenase) and HRPT1
(hypoxanthine phosphoribosyltransferase 1) were used as endogenous controls. Fold change
relative to the expression of the sample genes and reference genes was calculated using a
normalized expression (ΔΔ(Ct)) method using CFX manager software (Bio-Rad). Results
were expressed as a percentage of gene expression normalized by the corresponding control
(cells treated with the medium alone). Evaluated genes for retinoic acid pathway were as
follows: RBP (retinol binding protein), RAR (retinoic acid receptor), and RXR (retinoid
X receptor). Evaluated genes for skin regeneration genes were: HB-EGF (heparin-binding
EGF-like growth factor) and HAS2 (hyaluronan synthase).
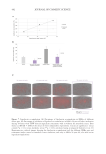
EVALUATION ON DERMIS REMODELLING: TYPE I COLLAGEN AND ELASTIN INDUCTION, AND
MATRIX METALLOPROTEASES 1 AND 3 (MMP-1 AND MMP-3) INHIBITION IN HUMAN DERMAL
FIBROBLASTS COCULTURED WITH HUMAN EPIDERMAL KERATINOCYTES
HEKa and HDFa were independently trypsinized and seeded in 12-well plates. The
cells in the coculture were incubated at 37°C in 5% CO
2 for 24 hours. After this
incubation period, the medium was removed, and fresh coculture medium was added
with testing products (S rebaudiana extract at 0.01% and retinoic acid at 0.05 µg/mL)
prepared in the same medium. Cells treated with coculture medium alone were used
as the coculture control. Cells were incubated for an additional 24 hours (for MMPs),
48 hours (for type I collagen), and 72 hours (for elastin), respectively, at 37°C in 5%
CO
2 humidified air. After the indicated times, supernatants were collected. A protease
inhibitor cocktail was added to the supernatants, and these were kept at -80°C until
they were analyzed by AlphaLISA (PerkinElmer, Inc., Waltham, MA, USA for type I
collagen), or ELISA (Cusabio, Houston, TX, USA, for elastin and ABCAM for MMP-1
and MMP-3) according to the manufacturer’s protocol. These results were normalized
with the total viable cell number for each condition calculated by the PrestoBlueTM
staining assay (Thermo Fisher Scientific, Waltham, MA, USA). The obtained values
were normalized compared to the coculture control and represent the means of three
independent experiments performed in duplicates. The statistical analysis used was the
unpaired student’s t test.
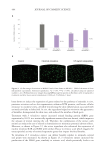
GENE EXPRESSION MODULATION ON HUMAN DERMAL FIBROBLASTS AND KERATINOCYTES
Keratinocytes or fibroblasts were seeded in a 24-well plate (NHEK) or 12-well plate
(NHDF) and then cultured in culture medium for 48 hours with the medium renewed
after 24 hours of incubation.
After this incubation time, cell culture medium was removed and replaced with the
medium containing or not containing (control) S rebaudiana extract, and cells were
incubated for 24 hours. All the experimental conditions were realized in n =3. At the end
of the incubation, culture supernatants were removed, and the cells were rinsed with a PBS
solution. The plates were immediately frozen dry at -80°C. Prior to RNA extraction, the
plates were thawed, and technical replicates were pooled. The total RNA of each sample
fluorescence intensity is proportional to the amount of the product in the PCR reaction.
Cycling conditions in the Bio-Rad CFX96 instrument are as follows: 95°C for 3 minutes,
followed by 40 cycles of denaturing at 95°C for 5 seconds, and annealing and elongation at
60°C for 30 seconds. GAPDH (Glyceraldehyde 3-phosphate dehydrogenase) and HRPT1
(hypoxanthine phosphoribosyltransferase 1) were used as endogenous controls. Fold change
relative to the expression of the sample genes and reference genes was calculated using a
normalized expression (ΔΔ(Ct)) method using CFX manager software (Bio-Rad). Results
were expressed as a percentage of gene expression normalized by the corresponding control
(cells treated with the medium alone). Evaluated genes for retinoic acid pathway were as
follows: RBP (retinol binding protein), RAR (retinoic acid receptor), and RXR (retinoid
X receptor). Evaluated genes for skin regeneration genes were: HB-EGF (heparin-binding
EGF-like growth factor) and HAS2 (hyaluronan synthase).
EVALUATION ON DERMIS REMODELLING: TYPE I COLLAGEN AND ELASTIN INDUCTION, AND
MATRIX METALLOPROTEASES 1 AND 3 (MMP-1 AND MMP-3) INHIBITION IN HUMAN DERMAL
FIBROBLASTS COCULTURED WITH HUMAN EPIDERMAL KERATINOCYTES
HEKa and HDFa were independently trypsinized and seeded in 12-well plates. The
cells in the coculture were incubated at 37°C in 5% CO
2 for 24 hours. After this
incubation period, the medium was removed, and fresh coculture medium was added
with testing products (S rebaudiana extract at 0.01% and retinoic acid at 0.05 µg/mL)
prepared in the same medium. Cells treated with coculture medium alone were used
as the coculture control. Cells were incubated for an additional 24 hours (for MMPs),
48 hours (for type I collagen), and 72 hours (for elastin), respectively, at 37°C in 5%
CO
2 humidified air. After the indicated times, supernatants were collected. A protease
inhibitor cocktail was added to the supernatants, and these were kept at -80°C until
they were analyzed by AlphaLISA (PerkinElmer, Inc., Waltham, MA, USA for type I
collagen), or ELISA (Cusabio, Houston, TX, USA, for elastin and ABCAM for MMP-1
and MMP-3) according to the manufacturer’s protocol. These results were normalized
with the total viable cell number for each condition calculated by the PrestoBlueTM
staining assay (Thermo Fisher Scientific, Waltham, MA, USA). The obtained values
were normalized compared to the coculture control and represent the means of three
independent experiments performed in duplicates. The statistical analysis used was the
unpaired student’s t test.
GENE EXPRESSION MODULATION ON HUMAN DERMAL FIBROBLASTS AND KERATINOCYTES
Keratinocytes or fibroblasts were seeded in a 24-well plate (NHEK) or 12-well plate
(NHDF) and then cultured in culture medium for 48 hours with the medium renewed
after 24 hours of incubation.
After this incubation time, cell culture medium was removed and replaced with the
medium containing or not containing (control) S rebaudiana extract, and cells were
incubated for 24 hours. All the experimental conditions were realized in n =3. At the end
of the incubation, culture supernatants were removed, and the cells were rinsed with a PBS
solution. The plates were immediately frozen dry at -80°C. Prior to RNA extraction, the
plates were thawed, and technical replicates were pooled. The total RNA of each sample