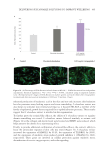
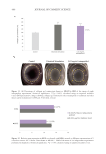
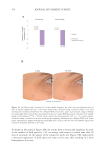
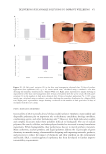
483
J. Cosmet. Sci., 75.5, 483–523 (September/October 2024)
*Address all correspondence to Donald S. Orth, orthconsulting@msn.com
The Evolution of Cosmetic Preservation and the
Microbiological Challenges Posed by Sustainability
DONALD S. ORTH,
Orth Consulting, LLC., Maineville, Ohio, USA
James L. Winkle College of Pharmacy, University of Cincinnati. Cincinnati, Ohio, USA
Accepted for publication July 8, 2024.
Synopsis
Cosmetic preservation has been evolving over the past 75 years. This review discusses changes in the use
of preservatives, including how the use of hurdle technology has enabled cosmetic scientists to formulate
products with reduced levels of preservatives or that are preservative-free, and how cosmetic preservation has
expanded from being focused just on preservative systems that prevent product contamination to products
that are sustainable because they preserve the environment and the healthy skin microbiome. There are still
gaps in our knowledge that need to be addressed to ensure that future products are adequately preserved, that
they do not encourage development of antibiotic-resistant microorganisms, and that they work with the skin
microbiome to maintain homeostasis.
INTRODUCTION
Cosmetic microbiology is a discipline within cosmetic science. A major objective of cosmetic
microbiology over the past 75 years has been to reduce or eliminate microbial contamination
of cosmetics in over the counter (OTC) drug products. This has been largely addressed by
preservative efficacy testing to demonstrate that formulas are adequately preserved for the
packaging and intended consumer use, by validating procedures used in the manufacturing
plant to maintain microbiological control, by finished product testing that is performed
correctly, and by use of appropriate microbiological release criteria to ensure that products
leaving the manufacturing plant are not contaminated with microorganisms that are able
to grow in the product.
A second goal of cosmetic microbiology has been to develop and use test methods that
are faster, more reliable, more environmentally friendly, and less expensive than the
microbiological test methods that have been used for decades. Such test methods expedite
product development and finished product release, with assurance that the test methods
are better able to recover stressed or metabolically injured microorganisms. This virtually
eliminates the growth of microorganisms in products that were initially tested and found
to meet microbial release specifications. Finding unacceptably high microbial counts in
J. Cosmet. Sci., 75.5, 483–523 (September/October 2024)
*Address all correspondence to Donald S. Orth, orthconsulting@msn.com
The Evolution of Cosmetic Preservation and the
Microbiological Challenges Posed by Sustainability
DONALD S. ORTH,
Orth Consulting, LLC., Maineville, Ohio, USA
James L. Winkle College of Pharmacy, University of Cincinnati. Cincinnati, Ohio, USA
Accepted for publication July 8, 2024.
Synopsis
Cosmetic preservation has been evolving over the past 75 years. This review discusses changes in the use
of preservatives, including how the use of hurdle technology has enabled cosmetic scientists to formulate
products with reduced levels of preservatives or that are preservative-free, and how cosmetic preservation has
expanded from being focused just on preservative systems that prevent product contamination to products
that are sustainable because they preserve the environment and the healthy skin microbiome. There are still
gaps in our knowledge that need to be addressed to ensure that future products are adequately preserved, that
they do not encourage development of antibiotic-resistant microorganisms, and that they work with the skin
microbiome to maintain homeostasis.
INTRODUCTION
Cosmetic microbiology is a discipline within cosmetic science. A major objective of cosmetic
microbiology over the past 75 years has been to reduce or eliminate microbial contamination
of cosmetics in over the counter (OTC) drug products. This has been largely addressed by
preservative efficacy testing to demonstrate that formulas are adequately preserved for the
packaging and intended consumer use, by validating procedures used in the manufacturing
plant to maintain microbiological control, by finished product testing that is performed
correctly, and by use of appropriate microbiological release criteria to ensure that products
leaving the manufacturing plant are not contaminated with microorganisms that are able
to grow in the product.
A second goal of cosmetic microbiology has been to develop and use test methods that
are faster, more reliable, more environmentally friendly, and less expensive than the
microbiological test methods that have been used for decades. Such test methods expedite
product development and finished product release, with assurance that the test methods
are better able to recover stressed or metabolically injured microorganisms. This virtually
eliminates the growth of microorganisms in products that were initially tested and found
to meet microbial release specifications. Finding unacceptably high microbial counts in