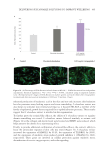
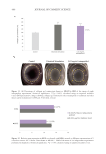
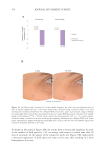
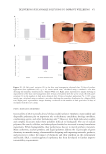
485 Evolution and Challenges of Sustainability
antimicrobial compounds), Jenner (created the smallpox vaccine—the world’s first vaccine),
Koch (developed Koch’s postulates—the four criteria used to establish a causal relationship
between a microbe and a disease), Pasteur (introduced pasteurization and disproved the
doctrine of spontaneous generation—the idea that living organisms could arise from
nonliving materials), Lister (promoted sterilization during surgery using carbolic acid, or
phenol, as an antiseptic), Bierjinek (founded virology and environmental microbiology), and
Winogradsky (initiated and developed microbial ecology).3
The contributions by these scientists and others resulted in the rapid growth of microbiology
in the latter half of the 19th century, and the time between 1980–1900 has been referred to
as the Golden Age of Microbiology because it was a time when the role of microorganisms
in fermentation was becoming known, and the microorganisms that caused many diseases
were isolated and identified.3 The Society of American Bacteriologists was founded in 1899.
This organization was the forerunner of the American Society for Microbiology, which
currently has nearly 40,000 members.
The deplorable conditions of the meat-packing industry in Chicago at the beginning of the
20th century were brought to light in The Jungle by Upton Sinclair. This book raised a public
outcry that resulted in passage of The Pure Food &Drug Act of 1906. This Act helped
ensure the sanitary manufacture of foods and drugs, required drug ingredient labeling,
and prohibited the sale of adulterated (e.g., contaminated) and misbranded products in
interstate commerce. The Food and Drug Administration (FDA) was established in 1931
however, it was not given regulatory authority for the safety of foods and cosmetics or for
the safety and efficacy of drugs. Visible mold growth was the primary microbiological
problem of products reported around this time.4 However, it is likely that many products
were contaminated with significant numbers of bacteria that were not detected because
products may not have had preservatives, and microbiological testing probably was not
done on a regular basis.
The time from 1900–1930 was a time of consolidation in which many investigators worked
to better understand the wealth of information generated during the Golden Age of
Microbiology. In the early 1900s, The American Public Health Association published the
first edition of Standard Methods for Examination of Water and Wastewater and forerunners of
Standard Methods for Examination of Dairy Products were published. The Digestive Ferments
Company (DifcoTM) was established in 1914. DifcoTM introduced dehydrated culture media,
which provided considerable time savings for media preparation as well as uniformity of
culture media used for growing bacteria, yeasts, and molds. Research on preservatives
began, and parabens became the preservative of choice because of their antibacterial and
antifungal activity.4
In 1937, the “Elixir of Sulfonamide” incident resulted in more than 100 deaths in the
United States due to consumption of an antibiotic, sulfonamide, in diethylene glycol that
was not safety tested. This and other tragedies resulted in enactment of the Food, Drug,
and Cosmetic Act of 1938, which gave the FDA responsibility for requiring manufacturers
to ensure safety—including the microbiological safety—of their products.3
EVOLUTION OF COSMETIC MICROBIOLOGY
In 1941, Maison deNavarre published The Chemistry and Manufacture of Cosmetics to provide
scientific information about cosmetic raw materials, formulations, and product preservation.5
antimicrobial compounds), Jenner (created the smallpox vaccine—the world’s first vaccine),
Koch (developed Koch’s postulates—the four criteria used to establish a causal relationship
between a microbe and a disease), Pasteur (introduced pasteurization and disproved the
doctrine of spontaneous generation—the idea that living organisms could arise from
nonliving materials), Lister (promoted sterilization during surgery using carbolic acid, or
phenol, as an antiseptic), Bierjinek (founded virology and environmental microbiology), and
Winogradsky (initiated and developed microbial ecology).3
The contributions by these scientists and others resulted in the rapid growth of microbiology
in the latter half of the 19th century, and the time between 1980–1900 has been referred to
as the Golden Age of Microbiology because it was a time when the role of microorganisms
in fermentation was becoming known, and the microorganisms that caused many diseases
were isolated and identified.3 The Society of American Bacteriologists was founded in 1899.
This organization was the forerunner of the American Society for Microbiology, which
currently has nearly 40,000 members.
The deplorable conditions of the meat-packing industry in Chicago at the beginning of the
20th century were brought to light in The Jungle by Upton Sinclair. This book raised a public
outcry that resulted in passage of The Pure Food &Drug Act of 1906. This Act helped
ensure the sanitary manufacture of foods and drugs, required drug ingredient labeling,
and prohibited the sale of adulterated (e.g., contaminated) and misbranded products in
interstate commerce. The Food and Drug Administration (FDA) was established in 1931
however, it was not given regulatory authority for the safety of foods and cosmetics or for
the safety and efficacy of drugs. Visible mold growth was the primary microbiological
problem of products reported around this time.4 However, it is likely that many products
were contaminated with significant numbers of bacteria that were not detected because
products may not have had preservatives, and microbiological testing probably was not
done on a regular basis.
The time from 1900–1930 was a time of consolidation in which many investigators worked
to better understand the wealth of information generated during the Golden Age of
Microbiology. In the early 1900s, The American Public Health Association published the
first edition of Standard Methods for Examination of Water and Wastewater and forerunners of
Standard Methods for Examination of Dairy Products were published. The Digestive Ferments
Company (DifcoTM) was established in 1914. DifcoTM introduced dehydrated culture media,
which provided considerable time savings for media preparation as well as uniformity of
culture media used for growing bacteria, yeasts, and molds. Research on preservatives
began, and parabens became the preservative of choice because of their antibacterial and
antifungal activity.4
In 1937, the “Elixir of Sulfonamide” incident resulted in more than 100 deaths in the
United States due to consumption of an antibiotic, sulfonamide, in diethylene glycol that
was not safety tested. This and other tragedies resulted in enactment of the Food, Drug,
and Cosmetic Act of 1938, which gave the FDA responsibility for requiring manufacturers
to ensure safety—including the microbiological safety—of their products.3
EVOLUTION OF COSMETIC MICROBIOLOGY
In 1941, Maison deNavarre published The Chemistry and Manufacture of Cosmetics to provide
scientific information about cosmetic raw materials, formulations, and product preservation.5