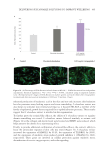
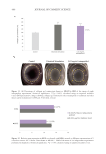
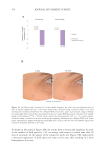
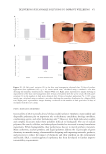
Table
II
Classes
of
Preservatives
with
Advantages
and
Disadvantages
Preservative
class
Advantages
Disadvantages
Organic
Acids
Benzoic Dehydroacetic
Salicylic
Sorbic
Good
activity
at
acid
pH
More
active
against
yeasts
and
molds.
Some
activity
against
bacteria.
Benzoates
are
used
in
carbonated
beverages.
Salicylic
acid
is
an
antimicrobial
active
ingredient
in
dandruff
and
acne
treatment
products.
Activity
is
pH
dependent
with
better
function
acidic
of
the
pKa:
Benzoic
acid
pKa
=
4.2
Dehydroacetic
acid
pKa
=
5.26
Sorbic
acid
pKa
=
4.76
Salicylic
acid
is
more
effective
with
pH
below
pH
3.7
(pKa
=
2.97)
Paraben
Esters
Methylparaben
Ethylparaben
Propylparaben
Butylparaben
Isobutylparaben
Active
against
Gram
positive
bacteria,
yeast,
and
mold.
Relatively
nonirritating
at
use
levels.
Some
esters
used
in
foods.
Not
effective
against
Gram-negative
bacteria
without
a
chelating
agent.
Inactivated
by
surfactants
above
the
critical
micelle
concentration.
More
effective
at
acid
pH.
Endocrine
issues
Possible
cancer
issues
Quaternary
Ammonium
Compounds
Benzalkonium
chloride
Benzethonium
chloride
Cetylpyridinium
chloride
Primarily
active
against
Gram
positive
bacteria.
Some
activity
against
Gram-negative
bacteria.
Incompatible
with
anionics
and
proteins.
Poor
activity
against
pseudomonads
without
chelating
agents.
More
active
above
pH
7
Formaldehyde
Donors
2-bromo-2-
nitropane-
1,3-diol
(BNPD)
Diazolidinyl
urea
DMDM
hydantoin
Imidazolidinyl
urea
Quaternium-15
Sodium
hydroxy-
methylglycinate
Broad
spectrum
of
antimicrobial
activity.
Donors
retain
activity
in
the
presence
of
surfactants.
Wide
pH
range
of
activity,
except
for
BNPD
which
is
not
stable
at
pH
6
Relatively
inexpensive
Possible
incompatibility
with
proteins.
Irritation/sensitization
issues.
Cancer
issues.
BNPD
use
has
decreased
since
the
1980s
due
to
the
potential
to
form
nitrosamines.
Regulatory
issues
(not
permitted
or
require
product
labeling
in
some
countries).
Alcohols
Ethyl
alcohol
Isopropyl
alcohol
Benzyl
alcohol 2,4-Dichlorobenzyl
Alcohol
Ethyl
and
Isopropyl
alcohols
have
broad
spectrum
of
antimicrobial
activity
at
high
concentrations
moderate
concentrations
(5%)
may
contribute
to
preservative
system.
Benzy
alcohol
is
antibacterial
and
antifungal
at
low
concentrations.
2,4-Dichlorobenzyl
alcohol
is
active
against
bacteria
and
viruses
and
is
used
in
throat
lozenges.
High
concentrations
(15%)
of
ethyl
alcohol
are
required
for
antimicrobial
action
(depending
on
system).
Ethyl
alcohol
is
volatile
and
may
evaporate
from
products
unless
this
is
prevented
by
packaging.
Alcohols
may
be
inactivated
by
nonionics.
Moderate
and
high
concentrations
may
cause
stinging
when
applied
to
skin.
Organic
Mercurials
Phenyl
mercuric
salts
Broad
spectrum
of
antimicrobial
activity
at
very
low
concentrations.
High
toxicity
and
irritancy.
Regulatory
issues
(allowed
in
eye-area
cosmetics
in
the
USA).
Inactivated
by
proteins
and
anionics
may
be
inactivated
by
nonionics.
Miscellaneous
Chloromethyl-
isothiazolinone
and Methylisothiazolinone Chlorphenesin
Antimicrobial
against
bacteria,
yeasts,
and
molds.
Isothiazolinones
are
effective
at
very
low
concentrations.
Chlorphenesin
is
relatively
nontoxic
and
is
approved
for
use
in
the
European
Union
(up
to
0.3%).
Isothiazolinones
may
cause
contact
dermatitis
and
are
inactivated
by
proteins,
high
pH,
and
bleach.
Phenoxyethanol
Rose
ether
fragrance.
Active
against
Gram-negative
bacteria
less
active
against
Gram
positive
bacteria.
Compatible
with
anionic
and
cationic
detergents
Inactivated
by
highly
ethoxylated
compounds
*Table
adapted
from
Orth. 14
489 Evolution and Challenges of Sustainability
II
Classes
of
Preservatives
with
Advantages
and
Disadvantages
Preservative
class
Advantages
Disadvantages
Organic
Acids
Benzoic Dehydroacetic
Salicylic
Sorbic
Good
activity
at
acid
pH
More
active
against
yeasts
and
molds.
Some
activity
against
bacteria.
Benzoates
are
used
in
carbonated
beverages.
Salicylic
acid
is
an
antimicrobial
active
ingredient
in
dandruff
and
acne
treatment
products.
Activity
is
pH
dependent
with
better
function
acidic
of
the
pKa:
Benzoic
acid
pKa
=
4.2
Dehydroacetic
acid
pKa
=
5.26
Sorbic
acid
pKa
=
4.76
Salicylic
acid
is
more
effective
with
pH
below
pH
3.7
(pKa
=
2.97)
Paraben
Esters
Methylparaben
Ethylparaben
Propylparaben
Butylparaben
Isobutylparaben
Active
against
Gram
positive
bacteria,
yeast,
and
mold.
Relatively
nonirritating
at
use
levels.
Some
esters
used
in
foods.
Not
effective
against
Gram-negative
bacteria
without
a
chelating
agent.
Inactivated
by
surfactants
above
the
critical
micelle
concentration.
More
effective
at
acid
pH.
Endocrine
issues
Possible
cancer
issues
Quaternary
Ammonium
Compounds
Benzalkonium
chloride
Benzethonium
chloride
Cetylpyridinium
chloride
Primarily
active
against
Gram
positive
bacteria.
Some
activity
against
Gram-negative
bacteria.
Incompatible
with
anionics
and
proteins.
Poor
activity
against
pseudomonads
without
chelating
agents.
More
active
above
pH
7
Formaldehyde
Donors
2-bromo-2-
nitropane-
1,3-diol
(BNPD)
Diazolidinyl
urea
DMDM
hydantoin
Imidazolidinyl
urea
Quaternium-15
Sodium
hydroxy-
methylglycinate
Broad
spectrum
of
antimicrobial
activity.
Donors
retain
activity
in
the
presence
of
surfactants.
Wide
pH
range
of
activity,
except
for
BNPD
which
is
not
stable
at
pH
6
Relatively
inexpensive
Possible
incompatibility
with
proteins.
Irritation/sensitization
issues.
Cancer
issues.
BNPD
use
has
decreased
since
the
1980s
due
to
the
potential
to
form
nitrosamines.
Regulatory
issues
(not
permitted
or
require
product
labeling
in
some
countries).
Alcohols
Ethyl
alcohol
Isopropyl
alcohol
Benzyl
alcohol 2,4-Dichlorobenzyl
Alcohol
Ethyl
and
Isopropyl
alcohols
have
broad
spectrum
of
antimicrobial
activity
at
high
concentrations
moderate
concentrations
(5%)
may
contribute
to
preservative
system.
Benzy
alcohol
is
antibacterial
and
antifungal
at
low
concentrations.
2,4-Dichlorobenzyl
alcohol
is
active
against
bacteria
and
viruses
and
is
used
in
throat
lozenges.
High
concentrations
(15%)
of
ethyl
alcohol
are
required
for
antimicrobial
action
(depending
on
system).
Ethyl
alcohol
is
volatile
and
may
evaporate
from
products
unless
this
is
prevented
by
packaging.
Alcohols
may
be
inactivated
by
nonionics.
Moderate
and
high
concentrations
may
cause
stinging
when
applied
to
skin.
Organic
Mercurials
Phenyl
mercuric
salts
Broad
spectrum
of
antimicrobial
activity
at
very
low
concentrations.
High
toxicity
and
irritancy.
Regulatory
issues
(allowed
in
eye-area
cosmetics
in
the
USA).
Inactivated
by
proteins
and
anionics
may
be
inactivated
by
nonionics.
Miscellaneous
Chloromethyl-
isothiazolinone
and Methylisothiazolinone Chlorphenesin
Antimicrobial
against
bacteria,
yeasts,
and
molds.
Isothiazolinones
are
effective
at
very
low
concentrations.
Chlorphenesin
is
relatively
nontoxic
and
is
approved
for
use
in
the
European
Union
(up
to
0.3%).
Isothiazolinones
may
cause
contact
dermatitis
and
are
inactivated
by
proteins,
high
pH,
and
bleach.
Phenoxyethanol
Rose
ether
fragrance.
Active
against
Gram-negative
bacteria
less
active
against
Gram
positive
bacteria.
Compatible
with
anionic
and
cationic
detergents
Inactivated
by
highly
ethoxylated
compounds
*Table
adapted
from
Orth. 14
489 Evolution and Challenges of Sustainability