345 Aging Skin Barrier
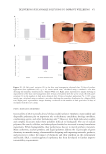
also reported that reaction to another strong irritant, octanoic acid was significantly lower
in subjects more than 55 years old while reactions to the weaker irritants acetic acid and
decanol were only directionally reduced with age.83 Bowman et al. performed a 10 day
cumulative irritation test comparing 26 subjects 18–45 with 26 subjects 65–80 using 11
products of widely varying irritancy.84 They found no statistically significant differences in
response between the age groups, but the response was directionally less in the older age
group for all the compounds that produced significant irritation.
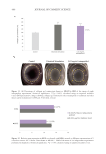
While the static barrier function of the epidermis does not seem to diminish with age,
recovery of barrier function after disruption has been found to be significantly slower in
older skin. Ghadially et al. disrupted SC by either tape stripping or acetone treatment to the
point that TEWL was increased to the range of 20–30 g/m2-hr.85 Recovery of the barrier
was quantified by measuring TEWL over the next several days. After acetone treatment
young skin (under age 30) recovered its barrier function by 50% at 24 hours, while older
skin (over 80) had only recovered 15% at 24 hours. Younger subjects took about 4 days
to recover to 90% of their original SC barrier to TEWL while older skin required 7 days
to recover by 90% after either tape stripping or acetone extraction. The slower recovery of
SC barrier function may be related to slower rates of epidermal renewal in older skin as
discussed below.
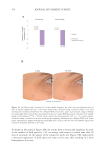
AGE AND SC TURNOVER
It is well known that corneocytes are continually shed into the environment and are being
continually replaced by cells generated in the SG (see above). The SC “turnover time” is
most frequently measured by staining the SC with the fluorescent dye, dansyl chloride, and
monitoring the rate of dye disappearance with a UV light. When all of the fluorescence
has disappeared the SC has turned over completely.86 Grove and coworkers reported that
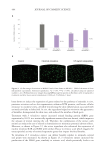
SC turnover time is increased in elderly skin.24,56 Data from taken from Grove et al. are
presented in Table II.
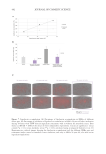
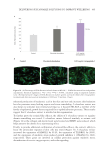
Figure 4. Scatter plot of TEWL versus age for 452 Chinese women along with a quadratic regression fi.
Figure courtesy of G.G. Hillebrand, used with permission.
also reported that reaction to another strong irritant, octanoic acid was significantly lower
in subjects more than 55 years old while reactions to the weaker irritants acetic acid and
decanol were only directionally reduced with age.83 Bowman et al. performed a 10 day
cumulative irritation test comparing 26 subjects 18–45 with 26 subjects 65–80 using 11
products of widely varying irritancy.84 They found no statistically significant differences in
response between the age groups, but the response was directionally less in the older age
group for all the compounds that produced significant irritation.
While the static barrier function of the epidermis does not seem to diminish with age,
recovery of barrier function after disruption has been found to be significantly slower in
older skin. Ghadially et al. disrupted SC by either tape stripping or acetone treatment to the
point that TEWL was increased to the range of 20–30 g/m2-hr.85 Recovery of the barrier
was quantified by measuring TEWL over the next several days. After acetone treatment
young skin (under age 30) recovered its barrier function by 50% at 24 hours, while older
skin (over 80) had only recovered 15% at 24 hours. Younger subjects took about 4 days
to recover to 90% of their original SC barrier to TEWL while older skin required 7 days
to recover by 90% after either tape stripping or acetone extraction. The slower recovery of
SC barrier function may be related to slower rates of epidermal renewal in older skin as
discussed below.
AGE AND SC TURNOVER
It is well known that corneocytes are continually shed into the environment and are being
continually replaced by cells generated in the SG (see above). The SC “turnover time” is
most frequently measured by staining the SC with the fluorescent dye, dansyl chloride, and
monitoring the rate of dye disappearance with a UV light. When all of the fluorescence
has disappeared the SC has turned over completely.86 Grove and coworkers reported that
SC turnover time is increased in elderly skin.24,56 Data from taken from Grove et al. are
presented in Table II.
Figure 4. Scatter plot of TEWL versus age for 452 Chinese women along with a quadratic regression fi.
Figure courtesy of G.G. Hillebrand, used with permission.