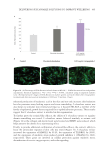
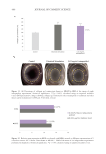
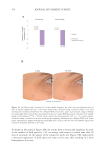
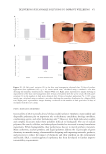
511 Evolution and Challenges of Sustainability
Currently, there is a great deal of research directed at studying the skin microbiome,
and some products involved with this technology have been introduced in recent years.
Sullivan et al. patented a skin treatment using Lactobacillus extract (i.e., a postbiotic) for
stimulation of β-defensins in skin cells to increase the skin’s natural defenses against
infection or to make products less irritating by decreasing the microbial density on
sensitive areas of the skin.160 A recent report by Spragge et al. indicated that the intestinal
microbiome protects against pathogens by “nutrient blocking” that is promoted by
diversity of the microflora and by the presence of key species that increase the overlap
between the nutrient use of the GI commensals and pathogens.161 It is believed that a
similar situation may occur on skin.
The intrinsic and extrinsic factors affecting the diverse microflora at the different skin
sites present a very complex system to study. Although single culture studies may be
informative, Boxberger et al. observed that there are clinical implications of the gut-brain-
skin connection in acne so that researchers may need to test with multiple microorganisms
under different conditions to better understand their full range of functions and how they
can be modified to lessen the severity of skin disorders and skin inflammation.162
The CNS may be candidates to be considered as probiotics on skin to lessen the severity of
acne, for wound management, and for down-regulation of the SIS. S. epidermidis, S. hominis,
and S. capitis are prominent members of the skin microbiome. They are able to produce
an array of antimicrobial substances (i.e., short-chain fatty acids from lipase hydrolysis of
sebum triglycerides, AMPs, and PSMs), and their ability to stimulate skin cells to produce
AMPs, including cathelicidins and defensins (to which they are refractory), make them
possible candidates to consider for use as probiotics. We are learning from studies of the
intestinal microbiome and the use of prebiotics. It is likely that selected ingredients will
act similarly and benefit desirable commensal microorganisms in the skin microbiome.
Finding a prebiotic (i.e., substrate) to facilitate growth or colonization resistance by selected
CNS in situ may be a better approach than trying to add living CNS for modulating the
skin microbiome to achieve desirable outcomes.
GAPS IN OUR KNOWLEDGE THAT NEED TO BE ADDRESSED IN FUTURE
PRODUCTS
The evolution of cosmetic preservation has involved transitioning from the use of traditional
preservatives to use of multifunctional ingredients to replace some or all the preservatives
used in some aqueous products. However, there are several gaps in our knowledge that
need to be addressed. These issues are discussed next.
ADEQUATE PRODUCT PRESERVATION
The goal of preservative efficacy testing is to determine the minimum concentration and
types of preservatives or multifunctional ingredients required for the adequate preservation
of aqueous cosmetic products. The acceptance criteria used for preservative efficacy testing
are critical, and more rigorous criteria provide a greater measure of protection than relaxed
criteria.
Unfortunately, there is no consensus on what is necessary or sufficient for preservative
efficacy test acceptance criteria. These criteria range from the rigorous (e.g., linear
Currently, there is a great deal of research directed at studying the skin microbiome,
and some products involved with this technology have been introduced in recent years.
Sullivan et al. patented a skin treatment using Lactobacillus extract (i.e., a postbiotic) for
stimulation of β-defensins in skin cells to increase the skin’s natural defenses against
infection or to make products less irritating by decreasing the microbial density on
sensitive areas of the skin.160 A recent report by Spragge et al. indicated that the intestinal
microbiome protects against pathogens by “nutrient blocking” that is promoted by
diversity of the microflora and by the presence of key species that increase the overlap
between the nutrient use of the GI commensals and pathogens.161 It is believed that a
similar situation may occur on skin.
The intrinsic and extrinsic factors affecting the diverse microflora at the different skin
sites present a very complex system to study. Although single culture studies may be
informative, Boxberger et al. observed that there are clinical implications of the gut-brain-
skin connection in acne so that researchers may need to test with multiple microorganisms
under different conditions to better understand their full range of functions and how they
can be modified to lessen the severity of skin disorders and skin inflammation.162
The CNS may be candidates to be considered as probiotics on skin to lessen the severity of
acne, for wound management, and for down-regulation of the SIS. S. epidermidis, S. hominis,
and S. capitis are prominent members of the skin microbiome. They are able to produce
an array of antimicrobial substances (i.e., short-chain fatty acids from lipase hydrolysis of
sebum triglycerides, AMPs, and PSMs), and their ability to stimulate skin cells to produce
AMPs, including cathelicidins and defensins (to which they are refractory), make them
possible candidates to consider for use as probiotics. We are learning from studies of the
intestinal microbiome and the use of prebiotics. It is likely that selected ingredients will
act similarly and benefit desirable commensal microorganisms in the skin microbiome.
Finding a prebiotic (i.e., substrate) to facilitate growth or colonization resistance by selected
CNS in situ may be a better approach than trying to add living CNS for modulating the
skin microbiome to achieve desirable outcomes.
GAPS IN OUR KNOWLEDGE THAT NEED TO BE ADDRESSED IN FUTURE
PRODUCTS
The evolution of cosmetic preservation has involved transitioning from the use of traditional
preservatives to use of multifunctional ingredients to replace some or all the preservatives
used in some aqueous products. However, there are several gaps in our knowledge that
need to be addressed. These issues are discussed next.
ADEQUATE PRODUCT PRESERVATION
The goal of preservative efficacy testing is to determine the minimum concentration and
types of preservatives or multifunctional ingredients required for the adequate preservation
of aqueous cosmetic products. The acceptance criteria used for preservative efficacy testing
are critical, and more rigorous criteria provide a greater measure of protection than relaxed
criteria.
Unfortunately, there is no consensus on what is necessary or sufficient for preservative
efficacy test acceptance criteria. These criteria range from the rigorous (e.g., linear