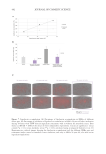
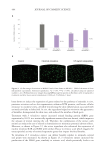
519 Evolution and Challenges of Sustainability
(86) Sokol J. Bright lights, big pity. Science. 2022 376(6591):340-343. doi:10.1126/science.abq4280
(87) Nakatsuji T, Kao MC, Zhang L, Zouboulis CC, Gallo RL, Huang C-M. Sebum free fatty acids enhance
the innate immune defense of human sebocytes by upregulating beta-defensin-2 expression. J Invest
Dermatol. 2010 130(4):985-994. doi:10.1038/jid.2009.384
(88) Dajnoki Z, Béke G, Kapitány A, et al. Sebaceous gland-rich skin is characterized by TSLP expression
and distinct immune surveillance which is disturbed in rosacea. J Invest Dermatol. 2017 137(5):1114-1125.
doi:10.1016/j.jid.2016.12.025
(89) Fischer CL, Drake DR, Dawson DV, Blanchette DR, Brogden KA, Wertz PW. Antibacterial activity
of sphingoid bases and fatty acids against Gram-positive and Gram-negative bacteria. Antimicrob Agents
Chemother. 2012 56(3):1157-1161. doi:10.1128/AAC.05151-11
(90) Béke G, Dajnoki Z, Kapitány A, et al. Immunotopographical differences of human skin. Front Immunol.
2018 9:424. doi:10.3389/fimmu.2018.00424
(91) Claesen J, Spagnolo JB, Ramos SF, et al. A Cutibacterium acnes antibiotic modulates human skin
microbiota composition in hair follicles. Sci Transl Med. 2020 12(570):12(570):eaay5445. doi:10.1126/
scitranslmed.aay5445
(92) Bitschar K, Sauer B, Focken J, et al. Lugdunin amplifies innate immune responses in the skin in synergy
with host- and microbiota-derived factors. Nat Commun. 2019 10(1):2730. doi:10.1038/s41467-019-10646-7
(93) Dessinioti C, Katsambas A. The microbiome and acne: perspectives for treatment. Dermatol Ther
(Heidelb). 2024 14(1):31-44. doi:10.1007/s13555-023-01079-8
(94) Christensen GJM, Scholz CFP, Enghild J, et al. Antagonism between Staphylococcus epidermidis
and Propionibacterium acnes and its genomic basis. BMC Genomics. 2016 17(1):152. doi:10.1186/
s12864-016-2489-5
(95) Wang Y, Kuo S, Shu M, et al. Staphylococcus epidermidis in the human skin microbiome mediates
fermentation to inhibit the growth of Propionibacterium acnes: implications of probiotics in acne
vulgaris. Appl Microbiol Biotechnol. 2014 98(1):411-424. doi:10.1007/s00253-013-5394-8
(96) Iwase T, Uehara Y, Shinji H, et al. Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm
formation and nasal colonization. Nature. 2010 May 20 465(7296):346-349. doi:10.1038/nature09074
(97) Coenye T, Spittaels K-J, Achermann Y. The role of biofilm formation in the pathogenesis and antimicrobial
susceptibility of Cutibacteriumacnes. Biofilm. 2022 4:100063. doi:10.1016/j.bioflm.2021.100063
(98) Smythe P, Wilkinson HN. The skin microbiome: current landscape and future opportunities. Int J Mol
Sci. 2023 24(4):1-28. doi:10.3390/ijms24043950
(99) Naik S, Bouladoux N, Linehan JL, et al. Commensal-dendritic-cell interaction specifies a unique
protective skin immune signature. Nature. 2015 520(7545):104-108. doi:10.1038/nature14052
(100) Lai Y, di Nardo A, Nakatsuji T, et al. Commensal bacteria regulate toll-like receptor 3-dependent
inflammation after skin injury. Nat Med. 2009 15(12):1377-1382. doi:10.1038/nm.2062
(101) Williams H, Campbell L, Crompton RA, et al. Microbial Host Interactions and Impaired Wound Healing
in Mice and Humans: Defining a Role for BD14 and NOD2. J Invest Dermatol. 2018 138(10):2264-2274.
doi:10.1016/j.jid.2018.04.014
(102) Lebre MC, van der Aar AMG, van Baarsen L, et al. Human keratinocytes express functional toll-like
receptor 3, 4, 5, and 9. Journal of Investigative Dermatology. 2007 127(2):331-341. doi:10.1038/sj.jid.5700530
(103) Wanke I, Steffen H, Christ C, et al. Skin commensals amplify the innate immune response to pathogens
by activation of distinct signaling pathways. J Invest Dermatol. 2011 131(2):382-390. doi:10.1038/
jid.2010.328
(104) Zhao C, Wang I, Lehrer RI. Widespread expression of beta-defensin hBD-1 in human secretory glands
and epithelial cells. FEBS Lett. 1996 396(2-3):319-322. doi:10.1016/0014-5793(96)01123-4
(105) Sørensen OE, Thapa DR, Rosenthal A, Liu L, Roberts AA, Ganz T. Differential regulation of beta-
defensin expression in human skin by microbial stimuli. J Immunol. 2005 174(8):4870-4879. doi:10.4049/
jimmunol.174.8.4870
(106) Grice EA, Segre JA. The skin microbiome. Nat Rev Microbiol. 2011 9(4):244-253. doi:10.1038/nrmicro2537
(86) Sokol J. Bright lights, big pity. Science. 2022 376(6591):340-343. doi:10.1126/science.abq4280
(87) Nakatsuji T, Kao MC, Zhang L, Zouboulis CC, Gallo RL, Huang C-M. Sebum free fatty acids enhance
the innate immune defense of human sebocytes by upregulating beta-defensin-2 expression. J Invest
Dermatol. 2010 130(4):985-994. doi:10.1038/jid.2009.384
(88) Dajnoki Z, Béke G, Kapitány A, et al. Sebaceous gland-rich skin is characterized by TSLP expression
and distinct immune surveillance which is disturbed in rosacea. J Invest Dermatol. 2017 137(5):1114-1125.
doi:10.1016/j.jid.2016.12.025
(89) Fischer CL, Drake DR, Dawson DV, Blanchette DR, Brogden KA, Wertz PW. Antibacterial activity
of sphingoid bases and fatty acids against Gram-positive and Gram-negative bacteria. Antimicrob Agents
Chemother. 2012 56(3):1157-1161. doi:10.1128/AAC.05151-11
(90) Béke G, Dajnoki Z, Kapitány A, et al. Immunotopographical differences of human skin. Front Immunol.
2018 9:424. doi:10.3389/fimmu.2018.00424
(91) Claesen J, Spagnolo JB, Ramos SF, et al. A Cutibacterium acnes antibiotic modulates human skin
microbiota composition in hair follicles. Sci Transl Med. 2020 12(570):12(570):eaay5445. doi:10.1126/
scitranslmed.aay5445
(92) Bitschar K, Sauer B, Focken J, et al. Lugdunin amplifies innate immune responses in the skin in synergy
with host- and microbiota-derived factors. Nat Commun. 2019 10(1):2730. doi:10.1038/s41467-019-10646-7
(93) Dessinioti C, Katsambas A. The microbiome and acne: perspectives for treatment. Dermatol Ther
(Heidelb). 2024 14(1):31-44. doi:10.1007/s13555-023-01079-8
(94) Christensen GJM, Scholz CFP, Enghild J, et al. Antagonism between Staphylococcus epidermidis
and Propionibacterium acnes and its genomic basis. BMC Genomics. 2016 17(1):152. doi:10.1186/
s12864-016-2489-5
(95) Wang Y, Kuo S, Shu M, et al. Staphylococcus epidermidis in the human skin microbiome mediates
fermentation to inhibit the growth of Propionibacterium acnes: implications of probiotics in acne
vulgaris. Appl Microbiol Biotechnol. 2014 98(1):411-424. doi:10.1007/s00253-013-5394-8
(96) Iwase T, Uehara Y, Shinji H, et al. Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm
formation and nasal colonization. Nature. 2010 May 20 465(7296):346-349. doi:10.1038/nature09074
(97) Coenye T, Spittaels K-J, Achermann Y. The role of biofilm formation in the pathogenesis and antimicrobial
susceptibility of Cutibacteriumacnes. Biofilm. 2022 4:100063. doi:10.1016/j.bioflm.2021.100063
(98) Smythe P, Wilkinson HN. The skin microbiome: current landscape and future opportunities. Int J Mol
Sci. 2023 24(4):1-28. doi:10.3390/ijms24043950
(99) Naik S, Bouladoux N, Linehan JL, et al. Commensal-dendritic-cell interaction specifies a unique
protective skin immune signature. Nature. 2015 520(7545):104-108. doi:10.1038/nature14052
(100) Lai Y, di Nardo A, Nakatsuji T, et al. Commensal bacteria regulate toll-like receptor 3-dependent
inflammation after skin injury. Nat Med. 2009 15(12):1377-1382. doi:10.1038/nm.2062
(101) Williams H, Campbell L, Crompton RA, et al. Microbial Host Interactions and Impaired Wound Healing
in Mice and Humans: Defining a Role for BD14 and NOD2. J Invest Dermatol. 2018 138(10):2264-2274.
doi:10.1016/j.jid.2018.04.014
(102) Lebre MC, van der Aar AMG, van Baarsen L, et al. Human keratinocytes express functional toll-like
receptor 3, 4, 5, and 9. Journal of Investigative Dermatology. 2007 127(2):331-341. doi:10.1038/sj.jid.5700530
(103) Wanke I, Steffen H, Christ C, et al. Skin commensals amplify the innate immune response to pathogens
by activation of distinct signaling pathways. J Invest Dermatol. 2011 131(2):382-390. doi:10.1038/
jid.2010.328
(104) Zhao C, Wang I, Lehrer RI. Widespread expression of beta-defensin hBD-1 in human secretory glands
and epithelial cells. FEBS Lett. 1996 396(2-3):319-322. doi:10.1016/0014-5793(96)01123-4
(105) Sørensen OE, Thapa DR, Rosenthal A, Liu L, Roberts AA, Ganz T. Differential regulation of beta-
defensin expression in human skin by microbial stimuli. J Immunol. 2005 174(8):4870-4879. doi:10.4049/
jimmunol.174.8.4870
(106) Grice EA, Segre JA. The skin microbiome. Nat Rev Microbiol. 2011 9(4):244-253. doi:10.1038/nrmicro2537