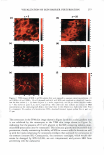
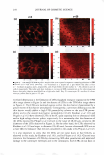
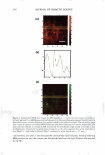
2008 ANNUAL SCIENTIFIC SEMINAR a Ceramide Fatty Acid No Fecal Extracts C I.! '"' :_------A _. -1 . ._. � - Ceramide 1-b----=================-IFatty Acid No Nasal Sttn:liou Ceramide El e e - - • Fatty Acid - • Ceramidase - .. Dragoderm Tea Extnct - Comrrey Lear - 355 Figure 1. Fecal extracts (a) and nasal secretions (b) are able to hydrolyze cerarnides. Selected cosmetic ingredients can inhibit ceramidase activity (c). Images are of thin layer chromatography plate containing samples of cerarnide incubated with biological sample or botanical in phosphate buffered saline, pH 7.4 for 18 hr at 25 °C. Biological exudates from humans can damage skin and result in skin irritation. Such irritation may be in part due to enzymatic degradation of vital skin components. Both fecal extracts and nasal secretions can degrade ceramide in vitro. When this degradation occurs in vivo, the reduction of this key stratum comeum lipid could eventuate in loss of skin barrier function and increase skin irritation. Table 1. Selected botanical compositions can change sphingomyelinase and ceramidase activity in vitro. Values are% change in activity observed as compared to control. buffered saline, pH 7.4 for 30 min at 25 °C. Botanical Dragoderm Aloe Ferox HS Phytoplenolin American Ginseng Tea Extract Comfrey Leaf Extract % Change in Sphingomyelinase Activity 450 410 430 320 -20 -30 Assays were conducted in phosphate % Change in Ceramidase Activity -90 -50 -50 -50 -90 -90 The in vitro results reported here indicate that botanical extracts can modulate the activity of enzymes normally involved in processing key skin lipids. Skin treatments that increase sphingomyelinase activity will produce ceramides, a key component of skin barrier function in vivo (4). Additionally, compositions that decrease ceramidase activity could preserve ceramide content in the stratum comeum. Taken together, coordinated control of ceramidase and sphingomyelinase in vivo may be possible with botanical compositions. If so, inclusion of these botanicals into products may present a cost effective means to deliver an important skin benefit. Literature Cited l. Kessner D, Ruettinger A, Kiselev MA, Wartewig S, Neubert RH. Properties of ceramides and their impact on the stratum comeum structure: part 2: stratum cornewn lipid models systems. Skin Pharmacol Physio/. 21(2): 58-74. 2008. 2. Choi MJ, Maibach HI. Role of ceramides in barrier function of healthy and diseased skin. Am J Clin Dermatol. 6(4): 215-223. 2005. 3. Machado M, Bronze MR, Ribeiro H. New cosmetic emulsions for dry skin. J Cosmet Dermarol. 6(4): 239-242. 2007. 4. Jensen JM, Schutze S, Neumann C, Proksch E. Impaired cutaneous permeability barrier function, skin hydration, and sphingomyelinase activity in keratin IO deficient mice. J Invest Dermato/. 115(4): 708-713. 2000.
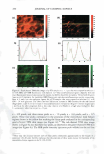
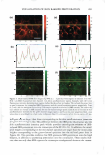
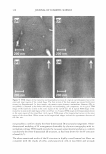
2008 ANNUAL SCIENTIFIC SEMINAR 357 Hematoxylin and eo in Cll. �El stainin& is a common technique used in histological analysis to examine the biological state of tissue. Image analysis was employed to separate the contribution of each stain, monitor stain intensity, measure the 2D area occupied by each stain, and count cell nuclei. One method for analyzing skin whitening or tanning is the Fontana-Masson stainine technique in which transverse sections of skin biopsies are examined for melanin expression. Using image analysis, me1anin content is quantified by measuring the total image area corresponding to melanin expression per length of skin as well as the density of the stain. See Figures 1 and 2. The Oil Red stainine experiment is often used to measure the amount of lipid matter in cell cultures. An application of this technique is to monitor the level of stored fat in adipocytes when treated with an active ingredient. Several image filters in conjunction with threshold techniques and image segmentation were utilized to quantify the amount of lipid present in the microscopic image. Immunonuorescent stainine techniques are commonly employed to monitor expression of certain cellular proteins. As an example, a green fluorescent dye, such as FITC or Alexa Fluor 488, is typically used to image the expression of Cytochrome C in fibroblasts. Using image algorithms, we demonstrate how the green fluorescence seen in the image can be quantified in terms of area per number of cells and fluorescence intensity. Other applications, using FITC or Alexa Fluor 488, were carried out in order to measure the expression of certain proteins in skin biopsies, such as Laminin V or Collagen IV located in the epidermal-dermal junction. Using image analysis techniques, we demonstrate how various image features, usually observed qualitatively, can be appropriately quantified. This was accomplished using standard methods of image analysis and applying them to images obtained from histological and cell culture analysis. References: [1] John C. Russ, The Image Processing Handbook, 4th Ed., CRC Press, 2002. [2] Y. Hu et al., J. lmmun. Meth,, 290: 93-105 (2005). [3] K. Matkowskyj et al., J. Histochem. Cytochem., 51: 205-214 (2003). Figure 1. Image of a skin section subjected to Fontana Masson staining. Figure 2. Segmentation of the image shown in Figure 1.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)