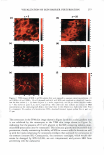
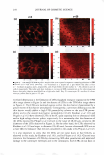
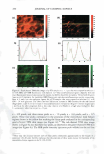
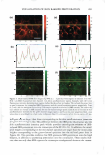
]. Cosmet. Sci., 59, 291-302 CTuly/August 2008) Microbiological assessment of the ultraviolet screening effect of sunscreen preparations YASMINE A. GOMAA, ELSAYED ABOULMAGD, NABILA A. BORAEI, INAS A. DAR WISH and LABIBA K. EL-KHORDAGUI, Departments of Pharmaceutics (Y.A.G., N.A.B., I.A.D., L.K.E.-K.) and Pharmaceutical Microbiology (E.A.), Faculty of Pharmacy, University of Alexandria, El-Khartoum Square, Azarita, Alexandria 215 21, Egypt. Accepted for publication February 11, 2008. This paper was presented as a poster in the Industrial Pharmacy section at the International Pharmaceutical Federation (PIP) Congress, Cairo, 2005. Synopsis The purpose of this study was the development of a microbiological method for the assessment of the ultraviolet (UV) screening effect of sunscreen preparations and determination of their sun protection factor. The method is based on the lethal effect of UV radiation on Escherichia coli (E. coli) and the protective ability of sunscreens. The time of UV exposure required for the reduction of the E. coli viable count by 90% (decimal reduction time, DRT) was used as the photoprotection assessment parameter. The method was tested by assessing the effect of selected experimental variables on the DRT. The suitability of the method as a quality control tool for sunscreen preparations was then checked by assessing the influence of selected formulation variables on the photoprotective effect of a series of o/w emulsion formulations with different compositions. The method proved valid for detecting changes in the photoprotective effect of a market sunscreen product as a result of modifying experimental conditions. It also proved valid for ranking market sunscreen products according to their UV screening effect. Equally important, the method could successfully detect changes in the photoprotective effect of sunscreen test formulations as a function of the concentration and type of the sunscreen agents. INTRODUCTION Determination of the photoprotective effect of sunscreen products is based on the as sessment of the photoprotection provided by such products against the erythemogenic effect of UVB radiation on human subjects. This test is performed according to the guidelines developed by the FDA (Food and Drug Administration) (1), COUP A (Eu ropean Cosmetic Toiletry and Perfumery Association) (2), the Australian Standard (3), and the Japanese Norm (4). Despite the many disadvantages of the in vivo methodology, Address all correspondence to Yasmine A. Gomaa. 291
292 JOURNAL OF COSMETIC SCIENCE such as ethical problems, fluctuation in radiation intensity, long irradiation times, and high costs, it remains the "golden standard" for sun protection factor (SPF) determina tion. During development of new sunscreen formulations, quick and inexpensive methods for estimation of UV screening performance are of great value. In vitro methods for SPF determination may serve this purpose. These include mainly spectrophotometric analysis of dilute solutions of sunscreen agents and determination of the transmission spectrum of thin films of sunscreen products (5). However, a shortcoming of in vitro methods is that they are based on the assumption that the UV protection provided by sunscreen products is merely due to the attenuation of UV radiation (UVA, UVB or both), according to the absorption characteristics and concentrations of the UV absorber(s) used (6). Further effects, which may be of relevance in vivo! are not taken into consideration. In the quest for a method combining the simplicity of in vitro methods and greater relevance to in vivo conditions, certain UV-sensitive microorganisms proved useful as living models. For instance, E. coli was utilized to model the effects of UV radiation on human skin (7). Further, Drosophila larvae were used to study the protective effect of sunscreens against UV-induced genotoxicity (8). The damaging effect of UV radiation on microorganisms (9,10) and the human skin (11-13) is initiated by photoalteration of DNA as a result of the formation of pyrimidine dimers. Replication of the UV-altered DNA is usually lethal to the microorganisms. Accordingly, microorganisms may play a role in the assessment of the damaging effect of UV radiation and, hence, the photoprotective effect provided by sunscreens. In a previous communication, the cidal effect of UV radiation on a certain strain of E. coli was investigated as an assay method for SPF determination of sunscreen products (7). The objective of this study was to develop a microbiological method for the assessment of the UV screening effect of sunscreen preparations and to assess the potentials of the method for the quality control and SPF prediction of sunscreen preparations. Develop ment of the method involved testing the influence of some experimental variables on the decimal reduction time (DRT). It also included testing the reproducibility of results and the sensitivity of the method using some market sunscreen products. The suitability of the method as a research and quality control tool was challenged by assessing the effect of some formulation variables such as sunscreen concentration and the incorporation of an additional sunscreen agent on the DR T. MATERIALS AND METHODS MATERIALS A standard strain of E.coli (NCTC* 10418) was used in the study. Luna® SPF 12, 27, and 35 (LUNA Cosmetics, Egypt), SpectraBAN® SPF 55 (Stiefel Laboratories Ltd, Sligo, Ireland), and Photoderm® SPF 100 (Laboratoire Bioderma, France) were the commercial sunscreen lotions tested. Benzophenone-3 (NEO HELIOPAN® BB) and phenylbenz imidazole sulphonic acid (NEO HELIOP AN® HYDRO/USP) were kindly provided by Symrise GmbH + Co. KG, Germany. Titanium dioxide, lanolin, white petrolatum, stearic acid, propylene glycol, triethanolamine, and edetate disodium were obtained * National Collection of Type Culture.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)