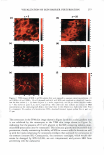
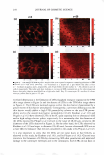
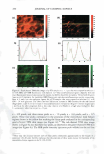
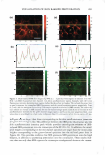
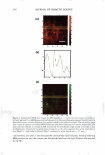
UV SCREENING EFFECT OF SUNSCREENS 301 time required for 50% rather than 90% reduction in viable bacterial count for Luna® products proved also to be linear (Figure 7). This time-saving modification may be appropriate for sunscreen preparations with a relatively high SPF. The microbiological method for assessment of photoprotection against UV radiation using E. coli as a test organism developed in this study may have several applications. As a research and quality control tool, the method is adequately sensitive to formulation variables that affect the protective effect of sunscreen formulations such as concentration, physicochemical interactions involving the active ingredient(s), and incorporation of additional sunscreen agents. Further, the method may be reliable as a simple, practical, and relatively inexpensive surrogate quality control tool for the routine evaluation of sunscreen preparations and prediction of their SPF values based on relationships obtained between the DR Ts and log SPF of market sunscreen products. Moreover, the method could be used to assess the effect of various factors on the viability of E. coli upon exposure to UV A or DVB radiation in the presence of factors capable of enhancing photoprotection or those inhibiting cellular damage such as antioxidants (21,22). .5 u .5 .,g u u Iii 'tl u -� Cl.I = ·= 60 .---------------------, 50 40 30 20 10 y = 96. 753x - 105.33 R2 = 0.9279 ■ 0 -•----.-----,-----.----�--�-- 1.05 1.15 1.25 1.35 log SPF 1.45 1.55 1.65 Figure 7. Relationship between log SPF of Luna® products and the time required for a 50% reduction in count.
302 JOURNAL OF COSMETIC SCIENCE CONCLUSION The microbiological method developed in the study may be considered an alternative method for photoprotective assessment that combines the greater reliability of a method using a living microorganism model and the simplicity and cost effectiveness of in vitro techniques. REFERENCES (1) U.S. Food and Drug Administration, Sunscreen drug products for over-the-counter human use: Final monograph, Federal Register, 64, 27666-27693 (1999). (2) COLIPA, Sun Protection Factor Test Method, Ref 94/289 (1994). (3) Australian/New Zealand Standard 2604, Standards Association of Australia, Sunscreen Products Evaluation and Classification (1998). (4) Japan Cosmetic Industry Association QCIA), Standard SPF Test Method, (1999). (5) G. A., Groves, P. P. Agin, and R. M. Sayre, In vitro and in vivo methods to define sunscreen protection, Austr.]. Derrnatol., 20, 112-119 (1979). (6) B. Herzog, S. Mongiat, K. Quass, and C. Deshayes, Prediction of sun protection factors and UVA parameters of sunscreens by using a calibrated step film model,]. Pharrn. Sci., 93, 1780-1795 (2004). (7) R. W. Jones, S. Smith, C. Boden, and B. G. Carpenter, A microbiological assay for the sun protection factor of sunscreen products,]. Pharrn. Pharrnacol., 50(Suppl. 9), S138 (1998). (8) M. Toyoshima, K. Hosoda, M. Hanamura, K. Okamoto, H. Kobayashi, and T. Negishi, Alternative methods to evaluate the photoprotective ability of sunscreen against photo-genotoxicity, Photochern. Photobiol. B, 73, 59-66 (2004). (9) A. J. Nataraj, J.C. Trent, and H. N. Ananthaswamy, p53 gene mutations and photocarcinogenesis, Photochern. Photobiol., 62, 218-230 (1995). (10) J. L. Zimmer and R. M. Slawson, Potential repair of Escherichia coli DNA following exposure to UV radiation from both medium-and low-pressure UV sources used in drinking water treatment, Appl. Env. Microbial., 68, 3293-3299 (2002). (11) B. S. Rosenstein and D. L. Mitchell, Action spectra for the induction of pyrimidine (6-4) pyrimidone photoproducts and cyclobutane pyrimidine dimers in normal human skin fibroblasts, Photochern. Pho tobiol., 45, 775-780 (1987). (12) J. Hildesheim, and A. J. Fornace, The dark side of light: The damaging effects of UV rays and the protective efforts of MAP kinase signaling in the epidermis, DNA Repair, 3, 567-580 (2004). (13) A. Besaratinia, T. W. Synold, H. Chen, C. Chang, B. Xi, A. D. Riggs, and G. P. Pfeifer, DNA lesions induced by UV Al and B radiation in human cells: Comparative analyses in the overall genome and in the p53 tumor suppressor gene, Proc. Natl. Acad. Sci. USA., 102, 10058-10063 (2005). (14) F. Modseen, J. D. Williams, and A. Secker, Standardization of inoculum size for disc susceptibility testing: A preliminary report of a spectrophotometric method,]. Antirnicrob. Chernother., 21, 439--443 (1988). (15) U. Citernesi, Photostability of sun filters complexed in phospholipids or [3-cyclodextrin, Cosrnet. Toiletr., 116, 77-78, 80-82, 84, 86 (2001). (16) D. L. Damian, G. M. Halliday, and R. S. Barnetson, Sun protection factor measurement of sunscreens is dependent on minimal erytherna dose, Br. J. Dermatol., 141, 502-507 (1999). (17) R. Wolf, B. Tuzun, and Y. Tuzun, Sunscreens, Derrnatologic Therapy, 14, 208-214 (2001). (18) W. Johncock. Sunscreen interactions in formulations, Cosmet. Toiletr., 114, 75-76, 78-82 (1999). (19) S. A. Wissing and R.H. Muller, A novel sunscreen system based on tocopherol acetate incorporated into solid lipid nanoparcicles, Int.]. Cosrnet. Sci., 23, 233-243 (2001). (20) J. Schulz, H. Hohenberg, F. Pflucker, E. Gartner, T. Will, S. Pfeiffer, R. Wepf, H. Gers-Barlag, and K. P. Wittern, Distribution of sunscreens on skin, Adv. Drug Deliv. Rev., 54(Suppl. 11), Sl57-Sl63 (2002). (21) F. Bohm, R. Edge, L. Lange, and T. G. Truscott, Enhanced protection of human cells against ultra violet light by antioxidant combinations involving dietary carotenoids,J. Photochern. Photobiol. B., 44, 211-215 (1998). (22) Y. O'Callaghan and N. O'Brien, The effect of carotenoids and tocopherols in the protection of human fibroblast cells against UVA-induced DNA damage, J. Dermatol. Sci., 34, 231-233 (2004).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)