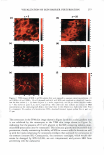
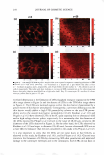
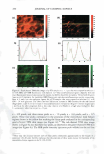
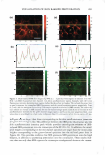
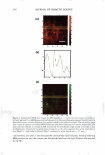
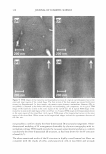
J. Cosmet. Sci., 59, 303-315 Quly/August 2008) True porosity measurement of hair: A new way to study hair damage mechanisms YIN Z. HESSEFORT, BRIANT. HOLLAND, and RICHARD W. CLOUD, Nalco Company, 1601 West Diehl Road, Naperville, IL 60563. Accepted for publication February 20, 2008. Presented at the Annual Scientific Seminar of the Society of Cosmetic Chemists, Anaheim, CA, May 11, 2007. Synopsis This study employs a novel method, gas sorption (1), to quantify the porosity characteristics of hair by determining total pore volume, adsorption pore-size distribution, and the surface area of damaged hair. Damage mechanisms were studied by comparing the different pore volume and surface area resulting from two different types of damage: chemical and UV. Hair color measurement and tensile strength, both reflecting the changes in hair cortex, were also employed in this study. The results suggest that hair damage caused by oxidative bleach and UV oxidation follows different pathways. Chemical damage (oxidative bleach) nearly triples the hair surface area in the first minute of bleaching due to the increase in the number of pores, followed by a sudden drop after 10 min of bleaching from smaller pores breaking down into larger ones. In contrast, UV damage shows an immediate loss in surface area in the first 200 hr of exposure and a gradual increase as exposure time continues. INTRODUCTION Many studies show that hair damage caused by chemical processes and UV exposure will result in increased hair swelling. Although much of the literature uses the terms "swell ing" and "porosity" interchangeably, most of the methods developed to determine hair damage are geared toward measuring swelling rather than porosity. The techniques used to measure swelling include water uptake (2), hair diameter change (3 ), liquid retention (4), and a centrifuge method (5). There has been no study to date revealing the details of pore size, pore volume and surface area that precisely defines hair damage through the measurement of hair porosity. Gas sorption, the method we have developed recently, enables us to quantify the different porosity characteristics of damaged hair (1). Fur thermore, we have undertaken research to study the damage mechanisms. Bleaching and sun exposure (UV) are considered two major causes of oxidative hair damage. Several studies have concluded that chemical oxidation follows S-S fission, which generates two moles of cysteic acid (6,7), while photochemical oxidation of cystine follows the C-S scission pathway (8), where only one mole of cysteic acid is produced. 303
304 JOURNAL OF COSMETIC SCIENCE The work of Ruetsch et al. (8) also concluded that melanin granules remain physically almost intact even after long-term UV irradiation/humidification. On the other hand, chemical oxidation (bleach) results in solubilization of melanin granules and loss of hair color. Their study further proves that the hair cuticle cells show extensive "thinning" and eventually "fuse" to the underlying cuticle layer after hair is exposed to the cycling of UV irradiation and humidification. Many investigations were also conducted to evaluate the deteriorated physical properties of damaged hair. The determination of the mechanical properties of degraded hair by bleaching and sun exposure illustrates that breaking strain and breaking stress are both affected by the two types of damage, but that chemical oxidation causes faster cortex degradation than photochemical oxidation (9). Several other analytical tools were also employed to examine hair damage, such as amino acid analysis, which exhibits a cor relation between the amount of amino acids and bleaching time (10), and Fourier transform infrared spectroscopy (FT-IR), which utilizes the ratio of sulfonate bonds (S=O) with the amide III band (11) to express the degree of hair damage. However, the majority of the studies have been centered either on the change in chemical composition of hair or a change in the mechanical properties of hair. The objective of this paper is to understand the difference in damage mechanisms between UV and chemical damage from a porosity perspective. Tensile strength and colorimetry analyses were also utilized to correlate porosity results. MATERIALS AND METHODS BLEACHING AND UV DAMAGE TREATMENT Virgin brown hair purchased from International Hair Importers Inc. was bleached by immersion in 6% hydrogen peroxide solution containing 1.7% ammonium hydroxide at 40 +/- 1 °C for 1, 5, 10, 15, or 20 min. UV EXPOSURE A QUV Accelerated Weathering Tester (Q-Panel Lab Products) with a 340 UVA bulb emitting at 340-nm maximum was used. The energy dose of the irradiation was 450 J/cm2 . Hair tresses of 1.5 g, each arranged in a single layer, were exposed under UV light for 200, 400, and 1200 hr. TENSILE STRENGTH The diameters of forty hair strands randomly selected from each group of chemical- and UV-damaged hair were measured using a fiber dimensional analysis system (Mitutoyo, Model LSM 5000). The hair samples were placed in a DiaStron miniature tensile tester (Model 170/670) for the determination of tensile strength in a wet condition. The total work force, normalized with the hair diameter, was calculated by using DiaStron soft ware (MTTWIN Application Software Version 5.0). The mean values obtained from 40 hair strands were analyzed using Tukey HSD statistical analysis to compare all the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)