458 JOURNAL OF COSMETIC SCIENCE
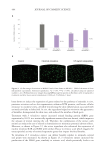
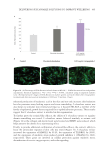
Analysis of the individual caffeoylquinic acid derivatives in the stevia extracts demonstrates
that these compounds are not well extracted by hot water or aqueous glycerin (Figure
4). Steric hindrance may contribute to the decreased extraction of the caffeoylquinic acid
derivatives with larger branched compounds extracting less with the hot water and aqueous
glycerin techniques. Extracting the 3,4-di-o-caffeoylquinic acid and 4,5-di-o-caffeoylquinic
compounds highlights the ability of subcritical water to better extract more complex
nonpolar compounds than hot water and aqueous glycerin, which is specifically 450 to
550 times more. The other caffeoylquinic acid derivatives have similar results. These data
demonstrate that subcritical water extracts caffeoylquinic acid derivatives from stevia at
efficacious levels that are not possible with hot water and aqueous glycerin.
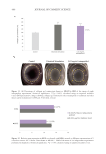
Figure 2. Changes in the polarity modify the behavior of water to extract a broader range of compounds.
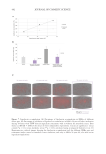
Figure 3. Comparison of extraction efficiencies of phytochemical families.
Analysis of the individual caffeoylquinic acid derivatives in the stevia extracts demonstrates
that these compounds are not well extracted by hot water or aqueous glycerin (Figure
4). Steric hindrance may contribute to the decreased extraction of the caffeoylquinic acid
derivatives with larger branched compounds extracting less with the hot water and aqueous
glycerin techniques. Extracting the 3,4-di-o-caffeoylquinic acid and 4,5-di-o-caffeoylquinic
compounds highlights the ability of subcritical water to better extract more complex
nonpolar compounds than hot water and aqueous glycerin, which is specifically 450 to
550 times more. The other caffeoylquinic acid derivatives have similar results. These data
demonstrate that subcritical water extracts caffeoylquinic acid derivatives from stevia at
efficacious levels that are not possible with hot water and aqueous glycerin.
Figure 2. Changes in the polarity modify the behavior of water to extract a broader range of compounds.
Figure 3. Comparison of extraction efficiencies of phytochemical families.