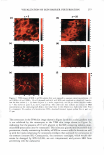
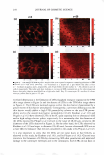
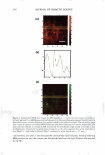
2008 ANNUAL SCIENTIFIC SEMINAR 367 First Screening f)EST C D I I I I I IDATES for punlig�Lprotection I •• • : : ... · � .. I •• • I I • � I I... \ I I: •• ."�'}·\. r1:" •••• _....-1'�--L._, • \ ·• . . .... .. . . . . .. .. . 240 2$0 260 270 280 290 300 310 320 330 3441 3IO 380 370 380 390 400 UV-c 5 Day Direct Sun Only - Delta a• Delta a from lnltl4l'�------ -- - 1+-----..--------- 5 ,.._p +-----'""'-c� ......... =---=--..-s 2 3 4 Day 5 .,._H Chart4a UV-a,, UV-a, Chart 3 5 Day Direct Sun Only - Delta E AE from Initial 7,.---- - - - -- - - - 6+--------- - - � - - 5 +---------�s,- -Notreatmer 4 ---A , -- P 3' i • H 2+--- - ��� i-s �1 - 2 3 Day 4 5 Chart4b
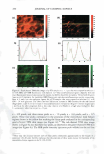
368 JOURNAL OF COSMETIC SCIENCE STRAIGHTFORWARD APPROACH FOR SCREENING SILICONE GELS OF VARYING FUNCTIONALITY FOR USE IN COSMETIC INGREDIENT DELIVERY Jeff Caruso, Summer L. Sivas, Ph.D., and Joan L. Meyer NuSil Silicone Technology, Carpinteria, CA 93013 Background: Biocompatibility of silicone gels makes them an excellent candidate for use as an encapsulant or matrix for delivery of active ingredients. Due to the myriad of silicone functionalities and even greater variety of cosmetic ingredients, a means to assess and screen for desired characteristics is required. Balancing formulation stability with acceptable delivery rate requires tailoring the matrix to the desired characteristics. � Rj CH, I 0-11 . o- 1 1 0-Si..l\/\/WVI CH3 CH3 D m Figure 1: Polysilioxane. R' = CH 3 , (CH2)CF3, (CH2)3O(CH2CH2O)0CH3 Silicones are derived from repeating silicon and oxygen atoms that create the polysiloxane chain. Figure I shows the generic structure of this polysiloxane chain. The R' side chain group represents various substituents that can be introduced onto the siloxane backbone. Incorporating different groups can directly influence the polymer's interactions with the surrounding environment. Methyl is the most common substituent group found in cosmetic formulations and biomedical applications. It is most known for its water resistance and desirable surface properties. Incorporating fluoro R' -groups can improve solvent resistance and modify surface properties while groups that are sufficiently hydrophilic, such as PEG-7, can make a polysiloxane water soluble. 1 In this study silicone gels varying in functionality from dimethicone, fluoromenthicone, and PEG-7 modified dimethicone are compared to demonstrate how variations in chemistry of the siloxane backbone can influence silicone's performance in cosmetic fluids. Water contact angle measurements and percent swell in several common cosmetic fluids are used to provide a straightforward screening method for the selection of suitable delivery matrices. Experimental: Silicone materials evaluated include: standard dimethicone (dimethyl) 100 molo/o trifluoropropyl methylsiloxane (fluoro) and 15 mo!% PEG-7 substituted dimethicone gels. Fluids evaluated include: water (H20), 50 mM phosphate buffer solution (PBS), mineral oil, cyclopentasiloxane (D5), methoxynonafluorobutane (CF-61), ethoxynonafluorobutane (CF-76), and 10 % glycolic acid (GA). Swell measurements were performed on all three gels with all the fluids listed above. Gel sample sizes were 0.5 g and allowed to swell at ambient for 48 hours. Fluid was decanted. Samples were then dried in an air-circulating oven to obtain the extracted sample mass for adjustment of final data. Time dependant Contact Angle (CA) measurements were conducted using Rame-Hart Model 200 and recorded with DROPimage software. Time dependant CA measurements were collected once per second for 180 seconds. Results and Discussion: Contact Angle. The time dependant H 2 0 CA for each silicone gel is plotted in Figure 2. CA is the measure of the angle (0) between the substrate surface and the line tangent form the droplet to the point of contact with the surface. CA is a sensitive enough measurement to characterize the nature of the material surface. A material that has a CA 90° is considered hydrophobic while 90° is characteristic of a hydrophilic surface. For absorbent materials, the CA continuously changes as a function of time where the water droplet penetrates or wets the surface. The dimethicone and fluoromethicone gels have contact angles greater than I 00° and a small rate of change over the time period tested. PEG-7 dimethicone has the lowest overall CA with a final CA of60 ± 5°. 1 Mark, J.E. Some Interesting Things About Polysilioxanes. Acc. Chem. Res., 37, 946-953, 2004.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)