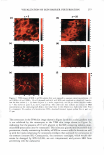
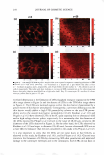
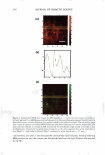
2008 ANNUAL SCIENTIFIC SEMINAR Time Dependant H,O Contact Angle $ 1eo 0 135 oo" 45° o+-····· - 0 30 80 90 Time(•) 120 --15mol'llPEO -Dimelh\,1 -Fluoro 100 Figure 2: Time dependant H2O Contact Angle of dimethicone, fluoromethicone, and PEG-7 modified dimethicone % Swell. Swell testing showed highest values for all samples with cyclopentasiloxane (D5), as high as 580 % for the dimethicone gel, see Figure 3. The lowest values occur with phosphate buffer ranging from 0-7 %. PEG-7 modified dimethicone retains affinity for nonpolar fluids, D5 and mineral oil, while u Co,melio Fluid EiTccLB: PEG 7 vs. Di1nclhioone 0 .J] n,o 10% CJA Mineral ll'A OU Coometk Fluid 05 •Dimethyl OPEG-7 Iii] IIJ CF-76 Figure 3: Measure of percent swell for dimethicone and PEG-7 dimethicone in cosmetic fluids Cosmetic Fluid Swell Effecl1' v.ith Fluorom�-thicone l'BS H,O Minenl Oil IPA Cosmetic Fluid D5 Figure 4: % Swell of fluoromethicone in cosmetic fluids Cli'-76 displaying increasing affinity for polar fluids, water and short-chained alcohols, compared with dimethicone. The presence of high ionic content (PBS) slightly reduces the degree of swelling compared to H20. Fluoromethicone demonstrates the lowest swell values for PBS, H20, mineral oil, and D5 compared to dimethicone and PEG-7 dimethicone, indicating the resistance to swell in many fluid types, see Figure 4. The largest percent swell for fluoromethicone was observed for CF-61 and CF-76, two fluoro-containing ether based solvents commonly used in a variety of cosmetic and skin care formulations. They are often used to replace or in conjunction with alcohol or other fast drying solvents. Accurate % swell values were not obtained for 10% GA with fluoromethicone. Conclusion: Contact angle and swell measurements are useful tools in initial screening of appropriate delivery matrices for cosmetic ingredients. The goal of the matrix delivered cosmeceutical device is shelf stability and an effective dosage delivery. Achieving these goals requires an understanding of the hydrophobic/ hydrophilic nature of the therapeutic compounds and silicones. Effective stability and delivery depends on the solubility and diffusivity of the compound in and through the silicone. The measurements performed provide an understanding of silicone interactions in various mediums. Based on these straightforward measurements one hopes to aid the formulator in selecting an appropriate starting point for their delivery matrix development needs. 369
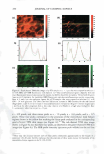
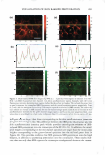
370 JOURNAL OF COSMETIC SCIENCE EXTENDED RELEASE OF LIDOCAINE FROM LINKER-BASED LECITHIN MICROEMULSIONS Jessica S. Yuan and Edgar J. Acosta University of Toronto Department of Chemical Engineering and Applied Chemistry Introduction The goal of transdennal drug delivery is to achieve adequate percutaneous absorption and permeation of active ingredients for local treatment. Microemulsions could improve skin absorption due to their large interfacial area and ultra-low interfacial tension. In our previous study (Yuan et al., 2008), non-toxic lecithin microemulsions were formulated using linker molecules with GRAS or food additive status. These formulations produce a significant increase in the absorption of lidocaine in the skin. In this work, we hypothesize that by topically administrating an active ingredient using the developed linker-based lecithin microemulsion, the active will be safely absorbed into the skin, thus producing an "in-situ" delivery patch for extended release. Potential advantages of this "in-situ" patch include its application on uneven and exposed parts, its low cost, and customizable dose. Traditional drug-in adhesive patches can not be used over bums, blisters, painful skin irritations or other skin wounds. Even for normal skin, they often cause skin irritation. The use of sprayable therapeutical microemulsions may represent a significant advance over the traditional patches. Since the microemulsions are transparent, the formulation could be used to treat various skin conditions without affecting the appearance of the patient. The objective of this study was to test the "in-situ" delivery patch hypothesis by evaluating skin absorption and release profile of lidocaine via linker-based microemulsions. Lidocaine-laden microemulsions were applied on pieces of cadaver pig ear skin and in vitro extended release studies were conducted. Moreover, fluorescent microscopy was used to study the permeation and location of a fluorescent dye within the skin strata. Experimental Microemu/sion preparation The method of preparing linker-based lecithin microemulsions and loading lidocaine was described elsewhere (Yuan et al., 2008). The microemulsions contain 4% lecithin, 12% sorbitan monooleate (hydrophobic linker), 1% (for Type II) and 7% (for Type I) sodium caprylate (hydrophilic linker), 3% caprylic acid, 0.9% sodium chloride, H20 and isopropyl myristate (1PM). The Type II formulation was loaded with 5 and 10% \idocaine, while the Type I formulation was loaded with 4 and 8 % lidocaine. In vitro extended release studies The in vitro extended release experiments were conducted in MatTek Permeation Devices (MPD). The pig ear skin was placed in a MPD with the epidermis facing up. A test formulation (0.4 ml) was applied in the donor compartment and was withdrawn after 30 min. The skin surface was blotted dry with an inert paper and was continued for extended release. The receptor compartment was filled with 5 ml of PBS. At predetermined times ( l , 3, 6, 12, 2 4 and 48 h), the receiver solution was withdrawn completely from the receptor compartment and was immediately replaced by fresh PBS solution. At 48 h, the experiment was terminated. Fluorescence Microscopy To visualize the "in-situ" patches, linker-based lecithin microemulsions containing 0.001% Nile red (fluorescent dye) were prepared and were used to conduct in vitro extended release. At predetermined times (1 and 6 h), the skin samples were taken off, blotted dry, and then rinsed twice with PBS. Solutions of 0.001 % Nile red in 1PM was considered as control. The skin samples were cross sectioned and were observed under a fluorescence stereomicroscope. Results and discussion Extended release from "in-situ" patches Fig. l reports the cumulative amount of lidocaine permeated across the skin as a function of time. For the "in situ" patches applying the Type II and I microemulsions, they typically show a large increase in the first 24h, followed by a continuous, slow increase up to 48h. Similar to the continuous dosage (Yuan et al., 2008), the one with the Type II fonnulations provided higher permeation amount than their Type I counterparts. When doubling the drug loading in either the Type II or I microemulsions, the lidocaine accumulation in the receiver also doubled. This result is in good agreement with the literature (Dreher et al., 1997) that transdermal drug delivery increases due to higher drug loading in microemulsions. The increased higher drug permeation from the "in-situ" patches is attributed to the larger lidocaine concentration absorbed in the skin after applying the microemulsions with higher drug loading. For example, by applying the Type II formulation with 5% lidocaine, the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)