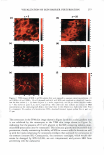
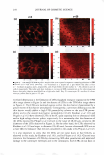
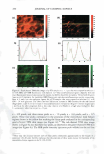
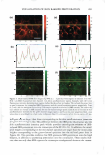
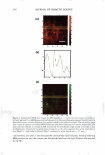
2008 ANNUAL SCIENTIFIC SEMINAR 371 lidocaine abso T tion in the skin was E 1000 490±19 µg/cm , while that for the � one with I 0% lidocaine doubled to � I 022±59 µg/cm2• j BOO During the extended release ,i experiments, most of the drug i 600 : absorbed in the skin was released. i Fig. 2 illustrates this observation by � plotting the fraction of lidocaine � rele ed as a function of time. In 1 200 the first 24 h, the drug release from I 0 • � • Type II -10% �Typtll-5% • A • Type I • 8% __.,_Typel-4% the "in-situ" patches applying the � Type I microemulsions is obviously u slower than those applying the 0 6 12 18 24 30 36 42 4i Tlme,h Type II formulations. Both of them Fig I. In vitro release profiles of lidocaine follow a first-order release with from "in-situ" patches via Type II and Type I different time constants of 0.04 h" 1 microemulsions with different drug loading. (for Type 11) and 0.03 h" 1 (for Type J). After 24 h, the Type I started to approach the fraction of lidocaine released of the Type II and eventually reached same. Microscopic observations Figures Ja-f show the location of the fluorescent dye (Nile Red) in the "in-situ" patches after applying the formulations of 1PM (Ja b), Type II (Jc-d) and J (Je-f) microemulsions at different time intervals. After 1 h (Fig. Ja, c and e), all formulations showed a deposition of Nile red in the superficial level of stratum comeum (SC) and a permeation in SC, the epidermis and dermis. In comparison to the 1PM system, both Type II and I microemulsions produced higher absorption of Nile red in the superficial layer and higher permeation in deeper skin layers. Especially, the Type II formulation had the highest deposition on the uppermost skin layer because its external phase is oil which contains more hydrophobic Nile red. As expected, the deposition in superficial level of SC clearly decreased after 6 h treatment (Fig. Jb, d and f). Because of the skin absorption, the microemulsions penetrated deeper layers and was able to diffuse uniformly further in the dermis to a great extent after 6 h application time. In contrast, the 1PM formulation did not 1.0 0.8 l Io.a I! ! j 0.4 · 0 ,, ::J 0.2 -�•Typell-10% -+-Type II - 5% ·A•Typel-8% -+-Type 1-4% 0.0 .,.: ---��-- r·-T--r-, 0 6 12 18 24 30 36 42 4i Tlme,h Fig 2. Fraction of lidocaine released from "in-situ" patches via Type II and I microemulsions with different drug loading �-- ... show the increase permeation of Nile red in deeper skin layers as the time of treatment increases. . ' ' - Conclusion We observed the extended release of lidocaine from the "in- (e) Type I, 1 h (f) Type I, Sh Fig. 3. Penetmtion of nile red into pig ear skin from (a) 1PM afu.."'1' lh, (b) 1PM after 6h, (c) Type TI situ" patches via linker-based lecithin microemulsions. After microemulsion after lh, (d) Type n microemulsion applying the microemulsions for a period of time, the drug was after 6h, (c) Type I microcmulsion after lh, (l) Type I absorbed in the skin which acted as drug reservoir and provided microemulsion oiler 6h extended release of the drug over 24 hours, with more than 99% released. Microscopic observations proved the drug uptake by the upper SC layer from the application of the linker microemulsions, and then the penetration into the deeper skin layers. In conclusion, linker microemulsions can act as "in-situ" delivery patches for extended release of active ingredients. Referenc Yuan et al., Linker-based lecithin microemulsions for transdermal delivery of lidocaine. International Journal of Pharmaceutics. 2008. 349:130-143. Dreher et al., Interaction of a lecithin microemulsion gel with human stratum comeum and its effect on transdennal Transport. 1997. Journal of Controlled Release, 45 (2): 131-140.
372 JOURNAL OF COSMETIC SCIENCE VESICULAR DELIVERY SYSTEMS: FROM PHOSPHOLIPIDS TO SILICONES FOR TARGETED SKIN SITES Shaow B. Lin, Ph.D.1, Stephanie Postiaux2, and Joanna Newton2, Ph.D. 1 Science & Technology, Specialty Chemicals Business, Dow Corning Corporation, Midland, MI 2 Application Development, Life Science Innovation Team, Dow Corning Europe, Seneffe, Befoium Email: shaow.lin@dowcorning.com Introduction: Vesicles are technically complex structure like "hollow particles" with aqueous phase at the exterior and the interior of the "particles", with a bilayer forming the vesicular particles. Because of this unique structure, vesicles are recognized as unique delivery systems for their ability to encapsulate lipophilic actives within the bilayer membrane. Some vesicles remain highly flexible and defonnable while loaded, making it possible to design vesicle that are capable of penetrating through the skin barrier layer and deliver actives to the target sites. Vesicles are historically synonymous to liposomes as vesicles are predominantly phospholipids-based. Recently a new class of silicone-based vesicles has been introduced and their potentials for delivery of actives have been suggested [1, 2). In this study, the structural characteristics of both phospholipid vesicles and silicone vesicles are described along with the comparison of their efficacy for the delivery of actives to targeted skin sites. Materials and Met.bods: The lipid-based vesicles in this report are prepared from soybean lecithin fraction which was further purified to high phosphatidylcholine (PC) and linoleic acid contents. The high PC/linoleic acid derived vesicles are flexible and very deformable, making them capable of penetrating into the upper skin layers. An ESR ( electron spin resonance) methodology was used in an ex-vivo evaluation to demonstrate the penetration of liposome containing model actives. Non-penetrating vesicular delivery systems consist of two structural types: sterically hindered liposomes and silicone vesicles. The sterically hindered liposomes are prepared with long PEG-grafted phospholipids on the exterior surface of the PC bilayer. Silicone vesicles are prepared from selected PEG-12 Dimethicone polymers via a Dow Coming patented process [3]. PEG-12 dimethicone is a rake-type linear silicone polyether with siloxane backbone and PEG-12 polyether grafted on the siloxane. Figure I illustrates (A) how the PEG-12 dimethicone arranges into bilayer and vesicular formation, and (B) structure of sterically hindered liposomes. (A) PEG polymer --. Dimethicone (B) Figure I. Non-penetrating vesicles options (A) Si vesicles from polymeric PEG-12 dimethicone, (B) sterically hindered liposomes from PEG-grafted phospholipid vesicles Results and Discussion: The vesicular delivery systems - both phospholipid-based and silicone-based vesicles, provided the essential protection to the encapsulated actives in aqueous cosmetic products, as have been previously demonstrated [2- 4].
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)