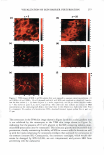
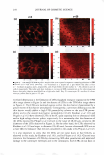
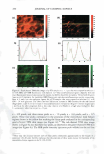
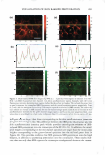
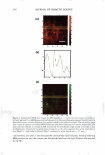
2008 ANNUAL SCIENTIFIC SEMINAR 373 The efficacy of delivery actives to the targeted skin sites is the focus of this study. The study of skin penetration of the encapsul ted actives and tbe liposomes are carried out using ESR (electron spin resonance) technique in ex-vivo with a test free radical. A formulation containing liposome encapsulated anti-oxidant was applied, and the radical scavenging activity within the epidermis layer was quantified over time to determine the antioxidant penetration efficacy through tbe upper layers of skin, compared to the natural scavenging properties of neat skin. The schematic of the ex-vivo experiment is illustrated in Figure 2. Neat Skin J 0 0 C Treated Skin \ 0 0 .,/ A�plicd fo:mulation /With anti-oxidants (blue) Stratum C•rneum Figure 2. Ex-vivo study on the penetration of liposome-encapsulated anti-oxidant through upper skin layer and scavenging of test radicals The non-penetrating vesicles, both silicone vesicles and liposomes, deposit encapsulated actives onto the surface of the skin (or target substrates). The encapsulated actives are released via mechanical rub-on after application and no penetration of the vesicular delivery systems into the skin are expected, based on the physical bulkiness or the sterically hindrance of the polymeric silicone molecules and the PEG-grafted phospholipids. Examples of actives for skin topical deposition such as vitamins, sunscreens, and plant extracts have been successfully encapsulated into these vesicles and further formulated into selected cosmetic formulations for such potential applications. Lipophilic actives such as vitamins, sunscreens, silicone fluid emollients, and hydrophilic actives such as vitamin C and natural extracts can be incorporated into Si vesicles via one of two Dow Coming proprietary process options: pre-load method or post-load method, where actives of interest are incorporated into pre-formed empty vesicles. It is important to note that the PEG-12 dimethicone for making Si vesicles is hydrophobic and exhibits poor to almost no solubility in water. Yet, the derived silicone vesicles show excellent bilayer flexibility, good active payload and good structural stability in aqueous environment. The Si vesicle is uniquely capable of delivering silicone fluid emollients at high payload, making it an ideal option for non-penetrating topical delivery applications. Conclusions: Two �damentally different vesicular technologies are compared for their fit for delivery of actives to targeted skin site applications. The skin penetration or non-penetration characteristic of liposomes is controlled at the molecular level, while both are capable of encapsulating and retaining lipophilic and hydrophilic actives. The PEG-12 Dimethicone derived silicone vesicles deposit lipophilic and hydrophilic actives onto the skin surface. References: I. Lin, S.B., Postiaux, S., Kim, G., "Development of A Novel Silicone-based Vesicle Delivery System," 23rcl IFSCC (Octo r 2004, Orlando, FL). 2. Lin, S.B., Postiaux, S., and Newton, J., "Novel Silicone Vesicles for Delivery of Actives in Personal Care," pp. 88-89, Program and Preprints of2005 Society of C05metic Chemists Annual Scientific Seminar (June 2005, Las Vegas, NV). 3. Lin, S.B., et al., WO2005103157, WO2005 l 03118, and WO2005 l 02248, assigned to Dow Corning Corporation (April 2005). 4. Blume, G., Sacher, M., Teichmuller, D., Jung, K., "Method/or The Analysis ofLiposomes," WO2007/003538Al (January 2007).
374 JOURNAL OF COSMETIC SCIENCE MIC ROEMULSIONS AS SPRAYABLE DELIVERY SYSTEMS FOR SPECIALTY INGREDIENTS Jonette Payne1, Terri Germain1, Gina Cosby1, Natalie Fasouliotu1 and Monna Manning2 1 Mclntyre Group, University Park, IL 2 Abitec Corporation, Columbus, OH Introduction Microemulsions are transparent and thennodynamically stable emulsions, meaning once they form they will remain stable unless "disturbed". The droplets are small enough (l0nm-lO0nm) that they do not interfere much with the passing of light and therefore appear clear (I 0nm-50nm) to translucent (50nm- l 00nm). Unlike Macroemulsions (400nm- 700nm), microemulsions fonn spontaneously without the need for a high input of energy and are therefore easy to prepare. When formulated properly, they are thermodynamically stable therefore they can have a long shelf life. They can also have a relatively low viscosity and do not require special handling equipment and lend themselves to spray applications as well as to traditional delivery systems. If oils or actives are the internal phase, the smaller droplet size offers greater area and more uniform application. 1 The size and curvature of the Surfactant/Emulsifier film at the Oil-Water interface determines the type of emulsion formed. Type I (O/W microemulsion) consist of swollen micelles surrounded by water where surfactant coexist with excess oil. Type II (W/O microemulsion)is the reverse of Type I where surfactants coexist with excess water. Type Ill consists of oil, water, and a bi continuous phase coexisting in a three phase equilibrium. Type IV is a single-phase microemulsion where both oil and water are completely solubilized in the surfactant microemulsion phase. 2 A flexible surfactant film is also important in fonning microemulsions. Depending on the molecular structure of the surfactant, certain co-solvents can improve the flexibility of the surfactant film (i.e. short chain alcohols), while other surfactant lend to a more rigid lamellar phase (Graph 2). Objective Since Type /Vmicroemulsions are single-phase, isotropic, and low-viscous fluids, they are particularly useful for cleansing and delivery of functional ingredients in skin care products.2 Many desired cosmetic active ingredients are not water soluble, e.g. organic sunscreens, tocopheryl acetate, essential oils and ester emollients to name a few. Products that incorporate these ingredients are typically opaque emulsions. Recently, clear sprayable products have been introduced, e.g. clear, sprayable sunscreens. However, these products contain a significant amount of short chain alcohols which can be drying to the skin. Microemulsion technology can be utilized to create a clear to translucent water-based active delivery system without the use of Ethanol or other short chain alcohols. Methodology Combine emollients with mid-range HLB emulsifiers to create an emulsifying concentrate via experimental design and evaluate its emulsifying potential. Emulsifiable ingredients tested include but are not limited to (with varying use levels depending on combination and design), Water Insoluble Vitamins, Fragrance Components, Silicones, Ester emollients, Essential oils, and Organic Sunscreens. The phase diagram (Graph I) is helpful when detennining the ratio of surfactant to co-surfactant when emulsifying a greater amount of"active" into a transparent microemulsion. 3 Results An emulsifying concentrate based on the low skin irritation ingredients PEG-6 Cafrylic/Capric Glycerides, Polyglycerol-6 Dioleate and Caprylic/Capric Glycerides was developed (Table I). Using 5-15% of the concentrate, we were able to create microemulsions of 1-3% of the tested ingredients (Table 2). Table I Microemulsion Blend3 IPEG-6 Caprylic/Capric Glycerides !Emulsifier HLB 12.5 80% IPolyglycerol-6 Dioleate !Emulsifier, humectant, emollient HLB 8.5 10% ICaprylic/Capric Glycerides ICo-Oil, Emollient Required 10% HLB 5-6
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)