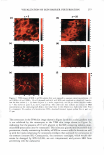
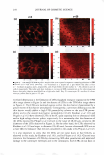
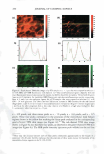
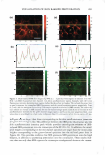
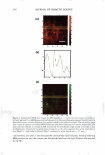
264 JOURNAL OF COSMETIC SCIENCE tants like sodium dodecyl sulfate (SDS) may penetrate into the skin and induce skin barrier perturbation by reducing the barrier properties of the stratum corneum (SC) (1-9). On the other hand, humectants like glycerol can maintain the water content of the skin and preserve the skin barrier (10-15). In addition, humectants like glycerol have been shown to mitigate surfactant-induced skin barrier perturbation in vitro (2). We have recently investigated the effect of SDS and glycerol on the skin barrier in vitro using macroscopic measurements of skin barrier perturbation in the context of a hindered transport aqueous porous pathway model (2). This analysis quantified the effect of glycerol on the SC morphology in vitro by showing that glycerol could reduce the size of the aqueous pores 1 in the SC relative to the size of the SDS micelles, such that the larger SDS micelle2 could not penetrate into the SC through the smaller aqueous pores and induce skin barrier perturbation. Because glycerol can mitigate SDS-induced skin barrier perturbation by modifying the SC morphology relative to: (a) a control (phos phate-buffered saline, PBS), (b) an aqueous solution of SDS, and (c) an aqueous solution of a mixture containing SDS and glycerol (2), one should be able to visually detect this change in SC morphology. Such a skin imaging capability can provide visual evidence of the ability of glycerol to mitigate SDS-induced skin barrier perturbation. In addition, once surfactants like SDS have penetrated into the SC, it is not known whether they are located in the keratins in the corneocytes or in the intercellular lipids in the lamellar bilayers comprising the SC. Several researchers have hypothesized that surfactants interact with the corneocyte keratins (6,9,26) and also with the lamellar lipid bilayers (5 ,28). A skin imaging technique, which can visualize the morphology of skin that has been exposed to an aqueous surfactant solution, may help shed light on this important issue. In addition, by contacting skin with surfactant solutions in the presence and in the absence of humectants, using an appropriate skin visualization technique, one can obtain fundamental insight into the modification of the skin barrier morphology induced by surfactants in the presence of humectants. Specifically, one may also be able to determine conclusively if a specific surfactant interacts strongly with the keratins in the corneocytes and/or with the intercellular lipids. Furthermore, one may also deter mine conclusively how such surfactant-skin interactions are modified by a humectant like glycerol when it is added to the aqueous surfactant solution contacting the skin. With the above need in mind, we have used two-photon fluorescence microscopy (TPM), an important invention in biological imaging (19), to visualize, as well as to quantify, the effects of surfactants and humectants on the skin morphology. Traditional biopsies of tissues, which are one of the principal pathological analysis methods for tissues such as human skin and pig full-thickness skin (p-FTS), can provide morphological infor mation with subcellular details. However, this method has some inherent limitations: (a) it involves tissue excision, fixation, and imaging to obtain useful morphological infor mation, and as such, is of an invasive nature, and (b) much of the cellular biochemical information is inevitably lost during the surgical and fixation procedures. On the other hand, advanced imaging methods like confocal laser scanning microscopy, 1 Structurally continuous, though tortuous, lacunar domains in the SC provide a morphological basis for the existence of aqueous pores in the SC (31-38). In fact, aqueous pores are the primary transport route for hydrophilic chemicals, such as an SDS micelle, to penetrate into, and across, a hydrophobic SC membrane (2,31-38). 2 Surfactants, such as SDS, consist of a hydrophilic head and a hydrophobic tail, and self-assemble to form micelles at a concentration above the critical micelle concentration (CMC) (3,4).
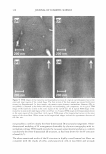
VISUALIZATION OF SKIN BARRIER PERTURBATION 265 which can address some of these limitations, are limited by: (a) a low penetration depth of scanning due to light-scattering effects and (b) accrued tissue sample photobleaching and photodamage due to repeated laser scanning (17, 18). In addition, most of the other imaging methods like electron microscopy (for example, scanning electron microscopy) can only achieve two-dimensional images that do not capture three-dimensional struc tures like blood capillaries and sebaceous glands in complex tissues such as the kera tinizing epithelium in human skin (19). TPM has overcome these diagnostic limitations (19). TPM is a non-invasive, three-dimensional imaging technique based on two-photon induced nonlinear excitations of fluorophores (16-19). It has the capability for deep tissue imaging (up to several hundred micrometers) and reduced photodamage, even for opaque and highly scattering tissues like human skin (16-20). In the past, studies have demonstrated the viability of using TPM to visualize, as well as to quantify, human skin morphological characteristics (20-23). In particular, Yu et al. developed a TPM visualization technique to visualize, as well as to quantify, the effect of a chemical enhancer, oleic acid (QA), on human skin (20-23). Specifically, Yu et al. used TPM to visually determine three-dimensional spatial distributions of fluorescent probe molecules, such as the hydrophilic probe sulforhodamine B (SRB), in excised full-thickness human cadaver skin exposed to PBS, the control, and to OA, the chemical enhancer. The fluorescent probe SRB was observed to reside primarily in the intercellular lipid bilayer region surrounding the corneocytes within the SC. In addition, from the TPM skin images, Yu et al. calculated the changes in the concentration gradient and in the vehicle-to-skin partition coefficient of the probe in the skin samples exposed to OA and to PBS (20). These calculations were subsequently used, along with theoretically derived mathematical expressions of transdermal transport, to quantitatively characterize the QA-induced relative changes to the skin-probe transport characteristics, including the SC diffusion coefficient and the SC diffusion length (20,23). These findings, utiliz ing the TPM skin imaging data analysis and transdermal transport modeling, demon strate that, in addition to providing visual scans that clearly delineate probe distribu tions in the SC, the subsequent quantification of these TPM skin images provides additional important insight into the mechanistic changes in transdermal transport underlying the visualized changes in probe distributions across the SC (20,23). In addition to the fluorescence originating from the hydrophilic fluorescent probes that have penetrated into the skin, the skin has an inherent florescence, referred to as the skin autofluorescence, that originates from endogenous fluorophores in the skin, including reduced pyridine nucleotides, flavoproteins, collagen, and elastin (24,25). Na et al. (25) showed that excitation wavelengths ranging from 340 to 380 nm, in the skin autofluo rescence emission spectrum, have two major component bands centered at 450 nm (75% of the spectrum) and 520 nm (25% of the spectrum). Using an appropriate filter set in the TPM apparatus, these emission wavelengths can be collected by the green channel, while a wavelength of 5 86 nm, which is the SRB emission peak, can be collected by the red channel (see the Experimental section). Since there is minimal wavelength overlap between the red and the green channels, a quantification of probe spatial distributions relative to the brick-and-mortar SC structure3 in the same skin sample, at precisely the 3 The stratum corneum (SC) has a brick-and-mortar structure, with the corneocytes as the bricks and the intercellular lipid bilayers as the mortar (44).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)