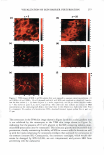
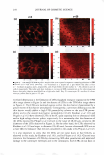
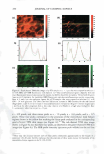
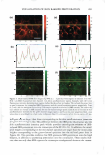
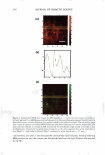
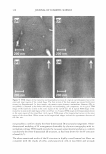
266 JOURNAL OF COSMETIC SCIENCE same skin spatial locations, can be achieved by this technique of dual-channel (the green channel and the red channel) TPM. Hence, the inherent skin structural features, as delineated in the green channel, can provide a fingerprint relative to the probe spatial distribution, as delineated in the red channel. Using dual-channel TPM, Yu et al. were able to show that OA drove the hydrophilic SRB probe to preferentially partition across the corneocyte envelopes and to diffuse into the corneocytes within the SC (21). We have used conventional, as well as dual-channel, TPM to visualize and quantify the effect of aqueous surfactant-humectant systems on p-FTS morphology4 relative to an aqueous control solution (PBS). Specifically, p-FTS samples were exposed to aqueous contacting solutions containing: (i) SOS (an anionic surfactant that is a harsh skin agent (2-6) (ii) SDS+glycerol (a surfactant/humectant mixture (2)) (iii) SCI (an anionic surfactant that is a mild skin agent (6,45)) (iv) PBS (the control) and (v) glycerol (a humectant that is a skin beneficial agent (10-15)). The TPM visualization studies were carried out using the hydrophilic fluorescent probe SRB, which penetrates into the skin through aqueous pores in the SC and hence, can shed light on the modification of these aqueous pores induced by surfactants in the presence of humectants (2-6, 20-23, 31-35, 38). Specifically, SRB emits a fluorescent signal in the red channel, has an emission peak wavelength of 586 nm, an absorbance peak wavelength of 565 nm, and a molecular weight of 559 Da (20-23). The p-FTS samples were exposed to aqueous contacting solutions i-v separately and, subsequently, were contacted with aqueous SRB solutions (see the Experimental section). Following these SRB exposures, the p-FTS samples were dried and visualized using the TPM apparatus to obtain conventional TPM skin images, as well as dual-channel TPM skin images. These TPM skin images were capable of determining the location of SDS within the SC, as well as the nature of the interactions of SOS with the SC. For example, if SDS penetrates into the corneocytes and denatures the corneocyte keratins, this will then result in increased corneocyte permeability to the fluorescent SRB probe. Therefore, the SRB probe will be located in the corneocyte domains of the SC and, accordingly, will be detected in the TPM skin images. Alter natively, if SDS penetrates into the intercellular lipid bilayers of the SC and disorders the lipid bilayer domains of the SC, then the SRB probe will also penetrate into the lipid bilayers and will be detected in the TPM skin images. The distribution of SRB in p-FTS samples upon contacting them separately with aqueous contacting solutions i-v was analyzed using a theoretical model presented in the Theoretical section. This analysis can shed light on the modification of the aqueous pores in the SC induced by aqueous contacting solutions i, ii, iii, and v, referred to hereafter as the enhancer, relative to aqueous contacting solution iv, the control. Finally, the results of the TPM visualization study, including the theoretical model results, are discussed in the Results and Discus sion section and summarized in the Conclusions section. EXPERIMENT AL MATERIALS Sodium dodecyl sulfate (SDS) was purchased from Sigma Chemicals (St. Louis, MO). Analytical-grade glycerol was purchased from VWR Chemicals (Cambridge, MA). 1 Because the permeability and morphological characteristics of p-FTS are similar to those of human skin, pig skin is an excellent model for human skin for both in vitro and in vivo skin permeability and imaging studies (26,46).
VISUALIZATION OF SKIN BARRIER PERTURBATION 267 Sulforhodamine B (SRB), a hydrophilic fluorescent probe, was obtained from Molecular Probes (Eugene, OR). Sodium cocoyl isethionate (SCI) from BASF was provided to us by Unilever (Edgewater, NJ). All these chemicals were used as received. Water was filtered using a Millipore Academic water filter (Bedford, MA). Phosphate-buffered saline (PBS) was prepared using PBS tablets from Sigma Chemicals and Millipore filtered water, such that a phosphate concentration of 0.01 M along with a NaCl concentration of 0.137 M were obtained at a pH of 7 .2 PREPARATION OF THE SOLUTIONS For the visualization of SRB in p-FTS, a solution of 0.05 mg/ml of SRB in PBS was prepared. The following aqueous solutions of surfactants, a humectant, and a surfactant+humectant mixture that contacted p-FTS were prepared: (i) a harsh surfactant solution-SOS (1 wt%) (ii) a harsh surfactant+humectant solution-SOS (1 wt%) + glycerol (10 wt%) (iii) a mild surfactant solution-SCI (1 wt%) 5 (iv) a control solu tion-PBS and (v) a humectant solution-glycerol (10 wt%). Note that a mild surfactant+humectant solution-SCI (1 wt%)+glycerol (10 wt%) did not induce skin morphological modifications different from those induced by solutions iv and v and, therefore, is not discussed here. PREPARATION OF THE SKIN SAMPLES Female Yorkshire pigs (40-45 kg) were purchased from local farms, and the skin (back) was harvested within one hour after sacrificing the animal. The subcutaneous fat was trimmed off using a razor blade, and the full-thickness pig skin was cut into small pieces (2 cm x 2 cm) and stored in a -80°C freezer for up to two months (2--4,34,45). IN VITRO DIFFUSION CELL SKIN EXPOSURE TO SURF ACT ANT-HUMECT ANT SYSTEMS FOLLOWED BY EXPOSURE TO SRB Prior to use in the TPM skin visualization experiments, the p-FTS sample was thawed for half an hour. The p-FTS sample was then mounted in a vertical Franz diffusion cell obtained from Permegear (Bethlehem, PA) with the SC facing the donor compartment. Both the donor and the receiver compart�ents were filled with PBS, and the p-FTS sample was left to hydrate for one hour to allow the skin initial barrier property to reach steady state. At this point, the skin electrical current across the p-FTS sample was measured at a 100 m V AC voltage (RMS) at 10 Hz, and only p-FTS samples with an initial skin current 3 µA were utilized in the permeation studies (2,45). The PBS in the donor compartment of the diffusion cell was then replaced with 1.5 ml of aqueous contacting solution i, ii, iii, or v. The aqueous solution in each donor compartment was then allowed to contact the p-FTS sample for five hours. Following this skin treatment with each aqueous contacting solution, the aqueous contacting solution was removed and 5 Note that p-FTS samples were contacted with aqueous SCI solutions at 35 ° C, while they were contacted with aqueous solutions i, ii, iv, and vat room temperature (25 ° C). This is because SCI (1 wt%) is soluble in water at 3 5 ° C, but not at 2 5 °C ( 6,45 ).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)