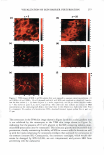
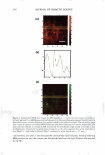
268 JOURNAL OF COSMETIC SCIENCE the donor compartment along with the p-FTS sample was rinsed four times with 2 ml of PBS to remove any trace chemical left on the skin surface and in the donor compart ment. Subsequently, each p-FTS sample was exposed to an aqueous SRB fluorescent probe solution in the diffusion cell for an additional 24 hours (20-23). Each p-FTS sample was then removed from the diffusion cell, rinsed four times with 2 ml of PBS as before, and blotted with a Kimwipe (Kimberly Clark, Roswell, GA) to ensure the removal of any excess SRB present on the skin surface. The circular area of the skin exposed to SRB was cut out with a surgical carbon steel razor blade (VWR Scientific, Media, PA), and subsequently sealed in a 2.5-mm imaging chamber (Cover well, Grace Bio-Laboratories, Bend, OR) with a coverslip (VWR Scientific) that con tacted the SC side of the p-FTS sample (20-23). A drop of PBS was added to the skin surface contacting the imaging chamber to prevent drying out of the p-FTS sample. The imaging chambers containing the p-FTS samples were imaged using the TPM appara tus, as described below. TWO-PHOTON MICROSCOPY (TPM) IMAGING OF THE SKIN SAMPLES The TPM imaging of the p-FTS samples was performed using the apparatus shown schematically in Figure 1 (20,29,30). This apparatus is based on a point-scanning approach, and has the capability for three-dimensional imaging. The incident beam is emitted by a femtosecond titanium-sapphire pulsed laser (Tsunami Spectra-Physics, Mountain View, CA) pumped by a 5W diode-pumped, solid state CW laser (Millenia V Spectra-Physics) (20,29,30). The two-photon excitation is provided at 780 nm and at a laser power of 200 mW (20). The incident beam generated by the laser is deflected by the X-Y scanner system, discussed elsewhere (30), to different angular positions, and then directed to the microscope system. The microscope system consists of a modified inverted microscope (Axiovert, lO0TV Ziess, Thornwood, NY) with an oil-immersion 40x objective lens (Zeiss F Fluar, NA .13) (30). To obtain dual-channel TPM images, Microscope System Skin Diode Laser Pumped Titanium-Sapphire X-Y "S..;...;.�"-"lscanner lane ! I I I I --------' ,---------------------------------------7 : �------� �-----� : •••·••··•• . .,,...,.,,...,,,.,..,.;,,-=:,,1 Photo Multiplier Photon 1 Tube Discriminator l I I I L ______________________________________ J Photon Detection-Conversion System Computer Figure 1. Schematic illustration of the apparatus used for the two-photon fluorescence microscopy (TPM) skin-imaging experiments (20,30).
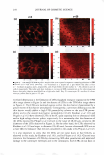
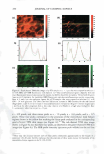
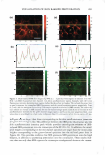
VISUALIZATION OF SKIN BARRIER PERTURBATION 269 the emitted photons from the laser excitation are collected by the microscope system and passed through a filter set consisting of a dichroic mirror with two filters (20). The filter set splits the signal into two: (a) the red channel for the photons emitted by the SRB fluorescent probe in the p-FTS sample and (b) the green channel for the photons emitted by the intrinsic skin fluorophores present in the p-FTS sample. The red and green channel signals are then sent to a photo multiplier tube (PMT), which along with the photon discriminator, comprise the photon detection-conversion system, shown in Fig ure 1. The output of the PMT is converted to single photoelectron pulses via the photon discriminator. The number of photons collected at each pixel, for the red and green channel images, is counted and recorded digitally by the computer (24,30). Aqueous contacting solutions i - (see Preparation of the Solutions section) were exposed separately to seven p-FTS samples, and six 100-µm x 100-µm skin sites were imaged for each p-FTS sample. Each p-FTS sample was imaged to a depth of 40 µm. Specifically, for each skin site imaged, 41 100-µm x 100-µm images were obtained, the first one at the skin surface (denoted as z = 0) and the last one 40 µm below the skin surface (denoted as z = 40), where z is the direction perpendicular to the surface of the skin. Each of the images was separated by a depth of 1 µm from the other. It was observed that most of the SRB probe was present in the p-FTS samples within a depth of 20 µm below the skin surface. Consequently, the TPM skin images obtained within the first 20 µm below the skin surface were analyzed (20-23), and images obtained at layers corresponding to z = 3 and z = 20 are presented in the Results and Discussion section. One should note that 3 µm below the skin· surface is within the SC, while 20 µm below the skin surface is within the viable epidermis (VE) (39,44). Therefore, the effects of aqueous contacting solutions i-v were visualized on the structure of both the SC and the VE (see the Results and Discussion section). DETERMINATION OF SC MORPHOLOGICAL CHANGES INDUCED BY SURFACTANT-HUMECTANT SYSTEMS USING A DECONSTRUCTION ANALYSIS OF THE DUAL-CHANNEL TPM IMAGES A recent investigation of the effects of aqueous SDS and aqueous SDS+glycerol contact ing solutions on the skin barrier has shown that the addition of 10 wt% glycerol to an SDS aqueous solution contacting the skin reduces the ability of the SDS micelles to penetrate into the epidermis, thereby significantly reducing the amount of SDS in the epidermis (2). Presumably, this reduction in the amount of SDS that can penetrate into the epidermis should also reduce the interactions of SDS with the keratins in the epidermis. Specifically, this should result in a reduction in the ability of SDS to rupture the corneocyte envelopes, and expose and disrupt secondary and tertiary structures of keratin in the corneocytes in the SC. On the other hand, mild surfactants like SCI are known to perturb the ordered intercellular lipid bilayers in the SC, thereby inducing skin dryness. In fact, researchers have hypothesized that the skin barrier perturbation response may be modulated by both of these two distinct mechanisms, that is: (a) the direct interaction of a penetrating skin agent with the keratins in the corneocytes in the SC and (b) bilayer disruption and delipidization of the SC lipid bilyers (5,6,9). Further more, either or both of these mechanisms can potentially trigger cytokine production, thereby triggering biochemical signals and promoting skin inflammation (5,9). There fore, in order to mitigate skin barrier perturbation induced by surfactant-humectant
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)