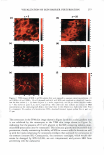
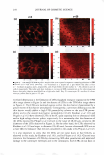
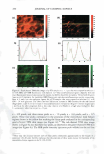
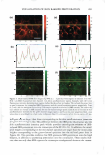
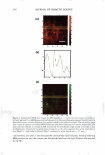
286 JOURNAL OF COSMETIC SCIENCE Table I Enhancements in the Slopes and Intercepts of the SRB Fluorescence Intensity Profiles as a Function of Skin Barrier Depth, and the Corresponding Theoretically Computed Aqueous Pore Characteristics (the Porosity-to-Tortuosity Ratio, si'r, and the Average Pore Radius, r p or ,) Induced by Aqueous Contacting Solutions i, ii, iii, and v Relative to Aqueous Contacting Solution iv, the Control Aqueous contacting solution E(slope) E(intercept) E(sh)* 1 p ore (i) 1 wt% SDS 6.5 ± 1.5 9.3 ± 0.9 6.5 ± 1.5 34 ± 5 (ii) 1 wt% SDS+ 10 wt% glycerol 3.1 ± 1.2 2.8 ± 0.7 3.1 ± 1.2 19 ± 6 (iii) 1 wt% SCI 2.5 ± 1.3 2.4 ± 0.6 2.5 ± 1.3 28 ± 5 (iv) PBS control 1 1 1 20 ± 3** (v) 10 wt% glycerol 0.5 ± 0.2 0.3 ± 0.2 0.5 ± 0.2 13 ± 5 * Note that E(slope) = E(e/'T), as discussed in the Theoretical section. ** This r p ore value, induced by aqueous contacting solution iv, was determined previously using log P-log R measurements in the context of a hindered-transport aqueous porous pathway model (2,34), and was utilized as an input to the model described in the Theoretical section to determine the r pore values induced by aqueous contacting solutions i, ii, iii, and v. iii, and v were determined, and are reported in Table I. 14 These rpore values are in excellent agreement with the rpore values obtained by us recently, using a different experimental/theoretical approach (2,45 ). The findings reported here indicate that an aqueous contacting solution of 1 wt% SDS (a harsh surfactant) induces the largest eh value relative to the other four aqueous contacting solutions considered, which indicates that SDS enhances skin penetration of SRB, relative to aqueous contacting solutions ii-v, through mode 2. Furthermore, an aqueous contacting solution of 1 wt% SCI (a mild surfactant) induces an rpore value that is closer to that induced by an aqueous contacting solution of 1 wt% SDS, while inducing a significantly smaller e/T value. This indicates that SCI is mild relative to SDS, because it reduces skin penetration of an irritant by reducing the eh of the aqueous pores without significantly reducing the average pore radius, relative to that induced by SDS. Table I shows that adding 10 wt% glycerol to a 1 wt% SDS aqueous contacting solution significantly reduces the rpore and eh values. Therefore, glycerol can mitigate SDS-induced skin barrier perturbation by closing aqueous pores through modes 1 and 2 (see the Theoretical section) such that SDS micelles (the irritant) cannot penetrate into the skin, a finding that is consistent with our recent study conducted using in vitro skin barrier perturbation measurements in the context of a hindered-transport aqueous porous pathway model (2). Finally, Table I also shows that an aqueous contacting solution of 10 wt% glycerol induces significantly smaller rpore and eh values than does the PBS control, thereby showing that glycerol preserves the skin barrier in vitro through a combination of modes 1 and 2. CONCLUSIONS The TPM skin visualization studies reported in this paper revealed that SDS induces corneocyte damage by rupturing corneocyte envelopes and denaturing keratins. 11 Note that the enhancement in the intercept induced by aqueous contacting solutions i-v relative to the control (aqueous contacting solution iv) is equal to the enhancement in the overall SRB partition coefficient, (E,) r (see the Theoretical section). In addition, note that the (E,) cl values were used to determine r pore ,B values by inputting r p ore,c = 20± 3 A (see the Theoretical section and reference 2).
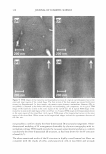
VISUALIZATION OF SKIN BARRIER PERTURBATION 287 This may further induce the creation of intra-corneocyte penetration pathways once SDS "opens-up" the cross-linked keratin structure of the corneocytes. Therefore, a group of such damaged adjacent corneocytes, taken together, may exhibit a large number of intra-corneocyte penetration pathways that may result in a localized transport region (LTR). A simultaneous quantitative analysis of the red- and the green-channel TPM skin images showed that an aqueous contacting solution of 1 wt% SDS + 10 wt% glycerol does not significantly induce corneocyte damage or L TR formation. Therefore, taken together with the results reported by us recently (2,45), these dual-channel TPM skin images provide additional evidence that adding 10 wt% glycerol to a 1 wt% SDS aqueous contacting solution significantly mitigates the ability of SDS to penetrate into the SC and interact with the keratins of the corneocytes and induce corneocyte damage. The dual-channel TPM images of p-FTS exposed to an aqueous 1 wt% SCI contacting solution showed a lower extent of SRB penetration into the corneocytes and into the intercellular lipid bilayers relative to the aqueous contacting solution of 1 wt% SDS. The PBS control solution induced localization of the SRB probe within the intercellular lipid bilayers surrounding the corneocytes of the SC. In addition, aqueous contacting solutions containing 10 wt% glycerol showed the least extent oflipid bilayer perturbation, and no effect on the corneocytes of the SC, relative to the other surfactant-humectant aqueous contacting solutions considered here. This important finding is consistent with in vitro skin barrier measurements reported by us in the literature (2,45), where an aqueous contacting solution of 10 wt% glycerol was shown to reduce the porosity-to-tortuosity ratio and the average radius of the aqueous pores relative to a PBS control. Therefore, these results indicate that glycerol, at an appropriate concentration, closes aqueous pores in the SC, which should reduce the extent of skin penetration of a hydrophilic irritant found in a skin cleansing formulation, such as a surfactant micelle, thereby mitigating skin barrier perturbation induced by the surfactant micelle. Consequently, cosmetic formulators may be able to use this mechanistic knowledge to tune the skin mildness of a formulation containing surfactants and humectants. For the five aqueous contacting solutions considered here, most of the SRB probe that penetrates into the skin barrier is present in the SC, and the probe intensity decays significantly as one visualizes the layers in the epidermis below the SC. We have quantified the amount of SRB that penetrated into the SC as a function of the SC depth upon contacting p-FTS separately with the five aqueous contacting solutions (i-v). This TPM analysis revealed that SDS enhances the probe partition coefficient the most, and that the extent of skin barrier perturbation induced by the five aqueous contacting solutions considered follows the order (from the highest to the lowest): i ii iii iv v. Therefore, a cosmetic formulator can use the TPM skin visualization and quan tification methodology presented here to screen new cosmetic formulations containing surfactants and humectants based on their ability to perturb the aqueous pores of the SC. The development of such an in vitro visual ranking methodology, including quantifica tion, can potentially reduce many costly and time-consuming in vivo human and animal testing procedures, thereby significantly reducing the cost and time-to-market of new cosmetic formulations containing surfactants and humectants. ACKNOWLEDGMENTS We thank Dr. Sidney Hornby and Dr. Yohini Appa from Neutrogena Corporation for useful discussions, and for providing partial financial support for this work.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)