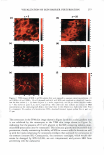
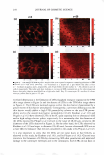
POROSITY MEASUREMENT IN HAIR 305 testing pairs (ANOVA one-way analysis of variance from JMP statistical software, SAS Institute, Cary, NC). HAIR COLOR MEASUREMENT Color co-ordinate L, a, b values for a hair tress were measured using a Hunter colorimeter (LabScan XE), where "L" stands for lightness, "a" for color of green to red, and "b" for blue to yellow. The total color change of LiE = [9*d12 + Lia2 + Lib2} 112 (12), in the case of hair damage by bleaching, was used to calculate the color difference between 0 min and 1 min bleaching or subsequently 1 min and 5 min, etc. The same calculation was also applied to UV damage. The total color change (LiE) represents the color difference between Ohr and 200 hr exposure, or 200 hr and 400 hr, until 1200 hr. We believe the LiE value for color change in this calculation clearly displays the progressive change of hair color due to the progressive damage from both bleach and UV. In addition, color change in the blue-yellow index (Lib) or the light index (Lil) was also included in the evaluation to further understand the degradation status of melanin granules after dam age. The average taken from six measurements in each tested tress represents the values of LiE, Lia, and Lil. GAS SORPTION Nitrogen adsorption studies on hair samples after bleaching and UV damage were conducted using a Quantachrome Autosorb-1 C instrument. Samples were cut in very fine pieces and then added to a sample cell where they were placed under vacuum at 145°C for 0.5 hr. Complete water removal is necessary to obtain accurate measurements, which is why 145°C was used. This value is based on the data collected from differential scanning calorimetry (DSC), in which a dehydration peak appears at about 125°C. A 5-pt BET (Brunauer-Emmett-Teller) surface area analysis was used for all samples. The total pore volume was collected at a P/P 0 of ~0.995. The pore size distribution curves are from the adsorption portion of the isotherm. RESULT AND DISCUSSION CHEMICAL DAMAGE Tensile strength analysis. The cuticle and cortex are the two main morphological compo nents in hair fibers. The cuticle is an important factor in torsional mechanical properties (13), but its contribution to bulk longitudinal mechanical strength is minor (14,15). The cortex constitutes the major part of the fiber mass of human hair and consists of elongated cortical cells, packed tightly and oriented parallel to the fiber axis. They contain microfibrils, which are composed of highly crystalline material, known as a helical proteins. It is believed that the cortex is primarily responsible for the tensile properties of human hair (15). The oxidation of cystine due to bleach causes the dis ruption of crosslinks, which results in the reduced wet tensile strength. Under our specific bleaching treatment, the wet hair tensile strength (Figure 1) displays a signif icant decrease in breaking force (about 20%) after the first minute of bleaching and another decrease in breaking force after 10 min. The statistical analysis (Figure 2) shows
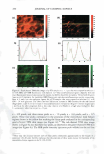
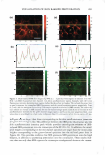
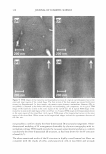
306 JOURNAL OF COSMETIC SCIENCE 2.5 0 -, Q) E 2 I:! - 0 · � :I: 1.5 0 � 1 s 1.306 1.442 1.118 1.188 1.05 0 0.5 0Mn 1 Mn 5Mn 10 Mn 15 Mn 20Mn Bleaching Time (min) Figure 1. Tensile strength of bleached hair. 0.0025 .L 0.0005 0 ........ -----.-------.-----.------,---�---........----------' 1 in 10 Min 15 Min 20Min 5Min Virgin Sample Name Figure 2. Statistical analysis for tensile strength measurement. All Pairs Tu key-Kramer 0.05 no difference between 1 min and 5 min of bleaching and also no difference between the pairs of 10 and 15, 10 and 20, and 15 and 20 min. It is understandable that the decrease in the wet tensile properties is primarily due to the degradation of disulfide bonds, but it is unclear why the change in tensile strength undergoes an immediate decrease in the first minute, and another after 10 min of bleaching time. Further analysis of color measurement and gas sorption was used to help find the answers. Hair color measurement. Hair pigments are distributed within the cortex and medulla in ovoid or spherical granules, and are not normally found in the hair cuticle of scalp hair. The oxidative attack on hair not only causes the breakage of disulfide bonds in hair protein, but it also breaks down the melanins. Wolfram and co-workers (16,17) have investigated the oxidation of human hair with and without pigment. Their research concluded that hair with pigment consumes hydrogen peroxide at a measurably faster rate than hair without pigment, suggesting a faster reaction of peroxide with hair pigment than with hair protein. Based on that conclusion, the initial response of oxi dative damage to hair fiber should be color change, under the assumption that the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)