258 JOURNAL OF COSMETIC SCIENCE Table I In-Shower Body Lotion Doses Calculated From Consumption Study Data Using the 15t\ SOt\ and 85th Percentile Adult Female Body Surface Area Data (13) Two-week study Five-week study Full body surface Reduced body surface Full body surface Reduced body surface Body surface area percentile 0.78 (0.045) 1.0 (0.057) 0.84 (0.026) 1.1 (0.033) 0. 71 (0.041) 0.90 (0.052) 0.75 (0.024) 0.96 (0.030) 0.62 (0.036) 0.80 (0.046) 0.66 (0.021) 0.85 (0.027) The calculations were performed using the full body surface area (arms, legs, full trunk) and a reduced body surface area (arms, legs, half-trunk) to obtain a more conservative dose estimate. Values reported are the mean calculated dose in µl/cm2 standard errors are shown in parentheses. performed for full (legs, arms, trunk) and reduced (legs, arms, half-trunk) body surface areas, the latter being done to provide a more conservative estimate of in-use dose even though habits and practices data indicate full body use in a high percentage of con sumers. Dose calculation results are summarized in Table I. Based on these results, an in-shower body lotion dose of 1 µl/cm2 was chosen for the controlled application pro tocol. This dose provides a number of uses from a commercial product bottle that is consistent with consumers' experience with the product (Table II) and is consistent with in-use doses reported for products of similar form that are applied to the body, e.g., sunscreens (14, 15 ). PROTOCOL PARAMETERS Wet time. An in-shower body lotion is applied to the skin after cleansing. Thus, the skin is wet for some period of time before the product is applied. A one-day (single appli cation) study was conducted to assess the effect of wet time on an in-shower body lotion product's potential to improve dry skin. After controlled cleansing, a water stream was run over the leg for 5, 10, or 15 minutes before in-shower body lotion application, to simulate in-use conditions. Evaluations were conducted one, three, and six hours after application. Wet time did not have a significant effect on the dry skin benefit resulting from in-shower body lotion application at any of these times (Figure 3). Wet time also Table II Predicted Number of Uses From a 250-ml Bottle of In-Shower Body Lotion for Various Doses Dose (µl/cm2) 2 3 4 5 6 Estimated number of uses from a 250-ml bottle 15th Percentile 50th Percentile 8 5th Percentile 20.4 18.5 16.2 10.2 9.2 8.1 6.8 6.2 5.4 5.1 4.6 4.1 4.1 3.7 3.2 3.4 3.1 2.7 Calculations were performed using the 15th, 50'\ and 85th percentile female body surface areas (13). Dosing at 1 µl/cm2 yields a projected number of uses that is consistent with consumer experience.
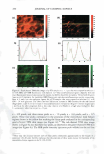
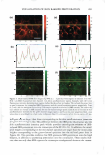
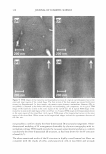
2.5 - 2.0 � :I 1.5 0 &&J .. en � +t _§ C IIJ II 1.0 Cl) • m E 0.5 0.0 DRY SKIN BENEFIT FROM IN-SHOWER BODY LOTION 259 P= 0.95 P= 0.89 -- - - - I•• - I•• 1 Hour 3 Hours Time After Treatment P= 0.18 - I•• - 6 Hours E � e .5 Cl) � � CJ c::::J 5 minutes 10 minu es - 15minutes Figure 3. Results from a single application study examining the effect of skin wetting time on visual dry skin improvement from an in-shower body lotion. Wet time did not significantly affect the dry skin benefit. had no significant effect on the in-shower body lotion's effect on stratum corneum hydration (p 0.13, data not shown). In-shower body lotion residence time and rinse time. A factorial study was conducted to examine the effect of residence and rinse times on the in-shower body lotion's potential to improve dry skin. Residence/rinse time combinations of 40 sec/10 sec, 40 sec/30 sec, 90 sec/10 sec, and 90 sec/30 sec were tested to cover the ranges identified in the in-shower body lotion habits and practices study. Skin condition was evaluated at baseline and three hours after the fifth treatment application. After five days neither residence time nor rinse time showed an impact (p 0.57) on the product's potential to improve d ry skin (Figure 4), or on its potential to improve stratum corneum hydration (p 0.17, data not shown). Petrolatum is the primary benefit agent in the in-shower body lotions tested, and the amount of petrolatum deposited on the skin during product use and remaining on the skin after rinsing is a key determinant of dry skin improvement efficacy. The results from this study indicate that that the amount of time the in-shower body lotion remains on the skin has a limited role in the benefit agent deposition process. This outcome is consistent with our experience with certain other rinse-off technologies that deliver petrolatum as a benefit agent, such as body washes. The apparent lack of an effect due to extended rinsing is not surprising for a hydrophobic benefit agent such as petrolatum the material tends to remain on the skin once deposited. DISCUSSION The studies conducted during protocol development show that the petrolatum depositing in-shower body lotion technology is robust in terms of its ability to provide dry skin benefits over a range of conditions that is relevant to how consumers shower and use an in-shower body lotion product. The final choice of protocol parameter values was driven by the need to maximize efficiency under clinical test conditions. Thus, the final
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)