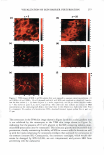
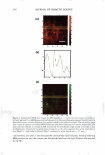
260 JOURNAL OF COSMETIC SCIENCE 2.0 C E 1.5 T T T 1 1 1 E :e -W U) U) U) +I 1.0 CD C C ca � a, CE C ·o 0.5 Q. "C C w 0.0 40 sec Residence 40 sec Residence 10 sec Rinse 30 sec Rinse 90 sec Residence 10 sec Rinse 90 sec Residence 30 sec Rinse Residence and Rinse Time Combinations Figure 4. Endpoint expert dryness evaluation (three hours after the fifth application) results from a study examining the effect of skin residence and skin rinsing times on dry skin improvement from an in-shower body lotion. A factorial analysis shows that neither factor has a significant effect on the observed dry skin improvement benefit (p 0.57). application procedure for testing the dry skin improvement potential of an in-shower body lotion is: • Subjects are recruited and their legs are washed with a non-moisturizing cleanser following a published procedure (12). • Wetting is continued for five minutes after rinsing the cleanser. • In-shower body lotion is applied at a dose of 1 µl/cm2 and rubbed on the skin for 20 seconds. • The in-shower body lotion remains on the skin for 40 seconds. • The skin is rinsed for 30 seconds and patted d ry . As noted earlier, two versions of in-shower body lotion were developed that deposit different amounts of petrolatum on the skin and deliver different levels of moisturization benefit. One important question is whether the protocol will discriminate among in shower body lotion products that are known to be different. A study was conducted to compare the two in-shower body lotion products and the control. The results, which are summarized in Figure 5, show that the protocol discriminates the in-shower body lotion products from each other and from the control, demonstrating the protocol's ability to show this product form's potential to provide a d ry skin benefit and to distinguish products formulated to provide different levels of dry skin benefit. A second important question is whether the protocol yields reproducible results. To test this, the same in-shower body lotion product was placed in studies conducted approxi mately one month apart. The results from this study, expressed relative to the control to account for changes in absolute scores, are summarized in Figure 6. These studies demonstrate the protocol's ability to reproducibly predict dry skin improvement po tential for an in-shower body lotion upon product retest.
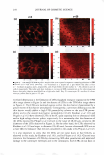
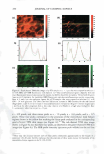
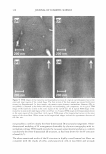
... C: Cl) E 1.5 � L. Q. E (I) 1.0 (I) - CD C � C C: C'G Cl 0.5 :E - ·sC: Q. "C C w 0.0 DRY SKIN BENEFIT FROM IN-SHOWER BODY LOTION 261 I I 1--··.. "-·· ·-· ...... · I Control (wash only) P0.01 P0.01 I P0.01 I In-Shower Body Lotion Medium Depositing I I In-Shower Body Lotion High Depositing ... ... +-' C: Cl) E g;? 0 a. E C: :i2 en � Cl I- ii � (!) Figure 5. Endpoint expert dryness evaluation (three hours after the fifth application) results from a study that compared two in-shower body lotion products formulated to deliver different levels of dry skin improvement. The protocol discriminates the products from each other and from the control. 2.0 .., :e IC] First Study Second Study I C w Cl) ti) E +I C: � 0 1.5 Cl) I■• E - g;? L. C Q. 0 e E u a. - E .E u, 0 1.0 C: (I) I■• :i2 Cl) .... en C B � �c Cl Cf I- Q) ),, ·-i 0.5 � = ,::, ns C (!) cii (U G) 0:: E 0.0 ...__ _ _._____.____. _____ ___.__...__..._____,_ ___ ____...___.__,_____...__-----L. 3 Hours 6 Hours 24 Hours Time After Fifth Treatment Figure 6. Expert dryness evaluation results following the fifth treatment in two five-day studies conducted with the same in-shower body lotion product. The tests were run approximately one month apart. The protocol developed to test in-shower body lotions yields reproducible results upon product retest.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)