BENZOYL PEROXIDE FORMULATIONS 535 priately diluted with the vehicle to achieve the desired dosage of 50 ml/kg by the oral route. Administration was accomplished with the aid of a 3-ml syringe adapted with a 20-gauge olive tipped needle. Each dose level was administered to 5 males and 5 fe- males. A separate group of 5 males and 5 females received the vehicle. The animals were observed for a period of 7 days for untoward signs and lethality and the LD,50 cal- culated by the Probit method (5). OCULAR IRRITATION EVALUATION New Zealand albino rabbits without regard to sex and weighing approximately 2 kg were obtained from a local supplier. • The animals were acclimated to laboratory condi- tions for 1 week prior to initiation of the test. The animals were maintained on Purina Chow and tap water provided ad libitum. Only animals judged free of ocular defects by macroscopic and biomicroscopic examination were utilized. The ocular irritation studies followed modifications of guidelines established by the Hazardous Substances Act. Procedures were modified by the addition of a 20-sec wash group. Three treatment groups of 6 eyes each were established for each test formula- tion. All groups received 0.1 ml of the test formulation in both eyes of each rabbit. Each eye was washed with a total of 300 ml of metered running tap water over a 2-min period. The time of eye washing after exposure to the formulation was different for each group. Group I eyes were washed at 20 sec after exposure Group II eyes were washed 5 min after exposure and Group III eyes were not washed until 24 h after ex- posure to the formulation. Appropriate sham control groups with washings on Day 0 and Day 1 were included for comparative purposes. A separate group of rabbits served as an untreated control. Sham control and untreated control groups were run each time the study was performed. After treatment, all eyes were examined at 1 h and at 1, 2, 3, 7, 14, and 21 days. All control groups of rabbit eyes were observed in conjunction with treatment animals. The conjunctiva was graded macroscopically by the method of Draize (6). The cornea, anterior chamber, iris, and lens were graded with the aid of a biomicroscope by the method of Baldwin, McDonald, and Beasley (7). Scores and inci- dences of each parameter were subjectively evaluated and used to rank the formula- tions either more or less irritating than similar competitor products. DERMAL IRRITATION EVALUATION Benzoyl peroxide formulations were tested in New Zealand albino rabbits of mixed sex and weighing about 2.5 kg. The animals were maintained on Purina Chow and tap water provided ad libitum. The backs of 15 rabbits were clipped free of hair and divided into 4 quadrants with each quadrant measuring approximately 2 in. on each side. The quadrants were labeled A, B, C, and D. Quadrants A and D in the first 6 rabbits were unabraded, while quadrants B and C of the same 6 rabbits were abraded with a Berkeley Scarifier.ñ Quadrants A and B of these first 6 rabbits received benzoyl peroxide 10 per cent, while quadrants C and D of •Krauss Rabbitry, Palestine, TX. -•Berkeley Biologics, Berkeley, CA.
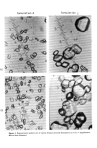
536 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS the same 6 rabbits received the competitive formulations. Quadrants A and B of the next 6 rabbits received the positive control, quadrants A, B, C, and D of 3 separate rab- bits served as untreated control quadrants. Each test formulation was applied at 0.5ml/dose, once a day for 3 consecutive days. After each application, the sites were covered with 3 x 3 in. gauze pads. The gauze pad was secured with an elastic adhesive bandage. At the end of each of the 3 24-h periods, the bandages were removed and dermal sites graded by the method of Draize (6). PHYSICAL MEASUREMENTS Particle size determinations were conducted using a Zeiss microscope • under standard 300 X magnification and 100 X magnification. Particle size determination was expressed as the per cent of particles which appeared within a certain micron range. The formulations studied were marketed products from several manufacturers. They consisted of some ingredients which were common to the formulations tested, i.e., A, B, C, and D (Table I). Table I Ingredients Common to Benzoyl Peroxide Formulations Evaluated Labeled as A, B, C, and D Benzoyl Peroxide Polyoxyethylene lauryl ether Alcohol Colloidal magnesium aluminum silicate Hydroxypropylmethyl cellulose Citric Acid Carbomer 940 Diisopropanolamine Disodium Edetate Dimethicone Acetone Propylene glycol Sodium lauryl sufate Fragrance H20 Triethanolamine RESULTS For purposes of this paper representative data will be presented only on formulation A among the best and formulation C as representative of the worst. Formulation D and B fell in between. *Carl Zeiss.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)