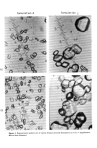
ELECTROSTATIC PROPERTIES OF HAIR 563 negative applied potentials, are plotted. The linear relationship between log V(t) and time in Fig. 9 confirms that the charge builds up and decays exponentially, as found by Wilson (8), Shashoua (9), and Ballou (10). We can characterize the process by the equations Q(t)=Qo[1- { tin2 exp - •77} ] (4) during charging, and Q(t)=Q0'exp{ tIn21 - (5) during discharging, where Q(t) is the charge on the hair at time t, Q0 is the equilibrium charge, r c is the half-life of charge induction, and r D is the half-life of charge decay. In this work, no systematic difference was found between values of r for positive and negative charges, nor between values for charging and discharging. This is shown by the superposition of the four curves at each humidity level in Fig. 9, and a similar result was obtained for all hair samples examined, at all humidity levels, whether treated with antistatic agents or not. We, therefore, computed the root mean square half-life as follows TI•,M S = 1/2 [(r+c) 2 + (T+D) 2 + (T_C) 2 -• (T_D)2] 1/2 (6) Our findings contrast with those of Shashoua (9), who found that r+ was generally not the same as r_ in textile fabrics and films, and that r c and r D were also unequal when the voltage exceeded 2000 V. Shashoua attributed the inequality off c and r D to ionization of the atmosphere. As discussed above, we found no such effect in this work. The logarithm of raMS varies linearly with relative humidity, over the range 20 to 50 per cent RH (Fig. 10). Shashoua (9) obtained a similar result for textile fibers and plastic films, over the range 15 to 65 per cent RH. Following this, we may write within these ranges TI•MS = T0 exp [-fi ß RH] (7) where -fi is the slope of the line, and r0 is the extrapolated half-life at zero per cent RH. Because of this linearity, experiments were routinely conducted at only two hu- midity levels (30 and 50 per cent) in this test also. Conventional shampoos and some creme rinses do not change r from its value for untreated hair other creme rinses reduce r significantly (Fig. 10). The level of agent remaining on the hair has a large effect on the half-life. The quaternary ammonium compound SBAC, when not rinsed off the hair before drying, reduces r to very low values. When it is rinsed off before drying, however, the half-life of the hair is little dif- ferent from that of untreated controls (Fig. 11). This is consistent with the findings of Sprokel (11) and Steiger (18), who showed that the surface conductivity of textile fabrics and yarns increases with the quantity of antistatic agents on the fibers. When the quaternary compound SBAC is rinsed off hair before drying, only small quantities remain on the fiber, so that the resistivity and hence the half-life are high.
564 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 10000 1000 100 10 SHAMPOO A UNTREATED • CREME A CREME RINSE B I I I i I 0 10 20 30 40 50 60 RELATIVE HUMIDITY, PERCENT Figure 10. Variation of charge mobility half-life TitMS of hair tresses with relative humidity. Charging potential: 2100 V
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)