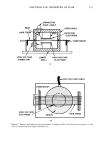
ELECTROSTATIC PROPERTIES OF HAIR 555 The test fixture, which we call here a Faraday shell, was constructed according to the ASTM description. The apparatus consists of 2 cylindrical brass shells, 10 cm in diameter. Each cell contains an electrode 5 cm in diameter which is insulated from ground by a polytetrafluoroethylene (PTFE) spacer. The electrodes are recessed 0.6 cm from the plane of the specimen. The walls of the fixture are grounded and are heavy to provide good electrical contact with the sample. The Faraday shell and the hair tresses under test were placed in a chamber of controlled relative humidity at 23øC. Safety interlocks, which disconnect the high voltage source, were contained in a box which covered the test fixture. This ensured that the high voltage supply could not de- liver a lethal shock to the operator. The hair tress was spread uniformly over a 3.8 cm width by inserting the tress at the plastic tab end in one of the combs mounted adjacent to the shells. A hand-held comb was inserted behind the fixed comb and pulled across the shell the tress was then affixed in the comb on the opposite side. The final position of the tress is shown in Fig. 2. The combs were used solely as a guide for specimen mounting, and were not used to generate charge on the tress. Any residual charges on the specimen after mounting were removed with a radioactive deionizer before closing the shells together. In addition to the provision of combs adjacent to the Faraday shell for mounting the specimen, the other important modification of the ASTM procedure was the partial en- closure of the bottom shell this shell was covered with a thin brass sheet with the ex- ception of a 4 cm width in which the specimen was mounted (Fig. 2). The purpose of this modification was to shield the detector electrode in the upper shell from stray fields of the charging electrode leaking around the specimen. In operation, a potential of--- 2100 V is applied to the electrode in the lower shell by a high voltage source at time zero. The upper detector electrode is used to monitor the electrostatic field potential, and is connected to an electrometer and chart recorder (see Fig. 3). At time zero, the detector electrode instantaneously charges by induction to a potential opposite to that of the charging electrode. Since hair is not a perfect conduc- tor, the charge initially induced on the hair is zero. Charge builds up on the hair by in- duction at a finite rate this charge has a polarity opposite to that of the high voltage source. Since the total charge within the Faraday shells is zero from Gauss' Law (15), the charge on the detector electrode decreases correspondingly. When the hair is fully charged, the charge on the detector electrode falls to zero. The output of the chart recorder during this process is shown in Fig. 4, together with a representation of the state of charge on the hair and on the two electrodes as the hair is charged and discharged. The charge, Q(t), on the hair at time t is related to the voltage, V(t), on the detector electrode as follows: Q(t) v(t) - 1--- Q0 v0 during charging, and Q(t) V(t) Qo Vo (2)
556 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (•) A ]D•'.I.'IOA i I
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)