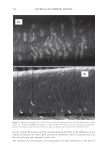
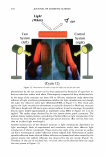
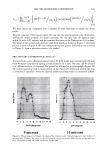
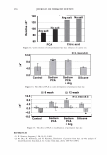
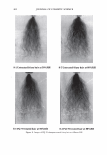
2006 TRI/PRINCETON CONFERENCE 351 Table lb Shampoos Used in Test II A B C D Control Sodium PCA Sodium PCA + Arg Silicone Na Laureth sulfate (active) 7.56 7.56 7.56 7.56 Cocamilopropyl betaine (active) 1.20 1.20 1.20 1.20 Sodium PCA 2.00 1.00 Arginine 0.40 Amodimethicone, emulsion 2.00 Pearlizing agent 4.00 4.00 4.00 4.00 Preservatives q.s q.s q.s q.s NaCl 2.00 2.00 2.00 2.00 Citric acid pH 5.8 pH 5.8 pH 5.8 pH 5.8 Water Balance Balance Balance Balance 10.0 a, 'i:" � ·- ca ra 5.0 +,I .c Q. I ::J .EJ en o L. 0.0 c( :::L 3 6 9 -5.0 pH value Figure 3. Arg uptake at various pH. Hair: Arg: 5.741 mM, 10 minutes immersion, 40°C. Arg Gly Ci) H3 N+ �� oW coo- Glu Hair pH 3 pH 6 pH 9 Figure 4. Ionic charge on amino acids and hair at various pH.
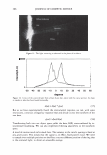
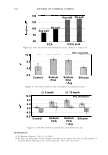
352 JOURNAL OF COSMETIC SCIENCE of the solution. It is also well known that the charges on hair also depend on the pH (Figure 4). The uptake of amino acids from the simple aqueous solutions is dominated by ionic interaction. Arginine, being a basic amino acid, bears cationic charge below pK 2 = 9.04, so has strong affinity for hair at pH over about 4, the isoelectric point of hair (6). Acidic or neutral amino acids having negative or neutral net charge at pH 3-7 are hardly taken up by hair. UPTAKE OF AMINO ACIDS FROM HAIR CONDITIONERS When we observe the uptake of amino acid from a cosmetic formulation, quite different phenomena occurs because cosmetic formulations are complex mixture of various chemi- cals. The example is shown in Figure 5. Although the interaction between glycine or glutamic acid and hair is very weak at pH 6, they are taken up by hair from conditioner formulations. This specific data was obtained from the material balance of the condi- tioner solution, so chemical interaction between the amino acids with the cationic surfactant probably makes the major contribution. USE OF ARGININE AS AN "ANCHOR" A guanidinium group is known to have quite high affinity for hair protein (7). Figure 6 shows the amount of arginine recovered from hair either by water or by acidic buffer solution (pH 3.5). The difference was greater at higher pH. This indicates the existence of strong interaction between acidic group on hair and arginine. This implies potential use of arginine as an "anchor" for the deposition of other cosmetic material with weaker affinity for hair. One example is application in combination with PCA. PCA is derived from glutamic acid in the human skin. Salts of PCA are highly hygroscopic and thus they are useful as moisturizers and conditioners in cosmetics. However, being an acid with small molecular size, its affinity to hair is relatively low. Figure 7 shows the uptake of PCA at various pH. The uptake rises at pH below 4. At pH over the pKa of PCA and hair, the uptake decreases because of the ionic repulsion between both carboxylic acids. When PCA is applied to hair as arginine salt, the uptake was larger than that of sodium salt (Figure 8). BENEFITS OF AMINO ACIDS FOR DYED HAIR Because of different chemical structure, amino acids are can provide a variety of functions :5" 1 00 ,-------------------, I so c, 60 a 40 .§. 20 -= 0 J! -201---- g -4o .__ __ A _ r ____ g G - ly ----- G lu __ ____, • Uptake from aqueous solutions (pH&) • Uptake from conditioner formulations Figure 5. Uptake of amino acids. Amino acid: 11.5 mM, 30 minutes immersion, 25 ° C.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)